Ch3cooh Lewis Structure - How To Discuss
 Daniel Cobb
Daniel Cobb 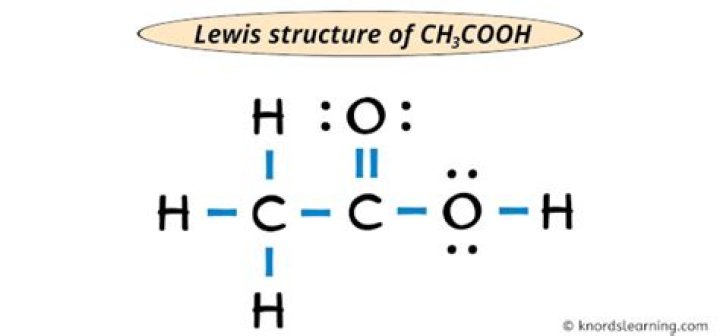
Ch3cooh Lewis Structure
Acetic acid, CH3COOH. What is the structure of Lewis, and have you explained geometry, ■■■■ hybridization ...?
Lewis structure:
Click on this page to view the structure:
...
Geometry:
Tetrahedral on the first carbon
While the triangular plane DI
Second carbon (with OH group)
Lewis structure of acetic acid
Acetic acid structure
Ch3cooh Lewis Structure
Ch3cooh Lewis Structure
Acetic acid, CH3COOH. What is the Lewis structure, and have you explained geometry, ■■■■ hybridization ...? 3
And the angle of attachment to the other carbon.
Lewis structure:
Click on this page to view the structure:
...
Geometry:
Tetrahedral on the first carbon
While the trigonal plane di
Second carbon (with OH group)
This page can help you.
D:
Acetic acid, CH3COOH. What is the Lewis structure, and have you explained geometry, ■■■■ hybridization ...?
And the angle of attachment to the other carbon.
Molecular geometry:
Depending on the carbon and hydrogen bonded oxygen (2 combined, 2 not combined)
Tetrahedron on carbon is bound to three hydrogens and another carbon (4 combined, 0 not combined)
Triangular planes on carbon, which are connected by double bonded oxygen, single bonded oxygen, and other carbons (3 combined, 0 not combined).
Electronic geometry:
On carbon, tetrahedron is bound to three hydrogens and another carbon.
Planar triangle on the other carbon.
In oxygen, tetrahedron combines with other hydrogen and carbon.
■■■■ hybridization:
For all hydrogen and oxygen: double bond s
For single bonded oxygen: SP
For carbon without hydrogen: SP 2 (as P square)
For carbon with hydrogen: SP 3 (looks like cubed P)
Since the hybridization around the other carbon is SP 2, the y triangle means a 120 degree separation.
I am not making a Lewis structure of acetic acid.
The geometry around C2 is trigonal-planar, hybridized with sp2 and the bond angle is 120.
Read the link



