Which equation shows the conservation of mass?
What equation shows the conservation of mass? (1) Na + Cl2> NaCl. (2) Al + Br2> AlBr3. (3) H2O> H + 02.
How is it shown that the mass is preserved?
In chemical reactions, mass is never lost or added. It is said that the mass is always preserved. In other words, the total mass of the products at the end of the reaction equals the total mass of the reactants at the beginning. This is because chemical reactions do not create or destroy an atom.
How is mass obtained in reactions?
This law says that despite chemical reactions or physical transformations, mass, that is, it can neither be created nor destroyed, is preserved in an isolated system. In other words, in a chemical reaction, the mass of the products is always equal to the mass of the reactants.
Which equation satisfies the law of conservation of mass?
N2 (g) + 3H2 (g) → 2NH3 (g) mass A balanced chemical equation satisfies the conservation law of mass. This law states that mass cannot be created or destroyed by any normal chemical or physical process. As you will recall, the number and type of atoms in a chemical reaction do not change.
What best describes the law of conservation of mass?
Explanation: The law of conservation of mass states that the mass of the reactive species is always equal to the mass of the species produced. So the mass cannot be created or destroyed, it can only be transformed from one place to another. Furthermore, the mass has no influence on the physical state of the reagents and products.
Does the mass change during a chemical reaction?
Mass cannot be created or destroyed in a chemical reaction. The law of conservation of mass states that the total mass of substances involved in a chemical reaction is conserved during the reaction.
Is the conservation of mass correct?
Given the mass-energy equivalence of relativity, the conservation of relativistic mass is the same as that of energy. The conservation of mass is therefore valid, so it should be noted that the mass of a system is not only the sum of the masses of the individual particles at rest, as is done conventionally.
Does the law apply to the maintenance of massive physical changes?
The law of conservation of mass states that mass is not created or destroyed by a chemical reaction or physical conversion. Therefore, the short answer to the question is yes, mass is conserved, regardless of whether matter is subject to chemical or physical changes.
Who made the conservation law?
Antoine Laurent Lavoisier
Why is the crowd held back in the comments?
Material cannot be created or destroyed by chemical reactions. It is the law of conservation of mass. In any chemical reaction, the products initiated into the reaction partners must have the same mass of matter. The balanced chemical equations show that chemical reactions maintain mass.
Why does mass reduce in a chemical reaction?
The mass of a chemical reaction is constant in a closed system. It is important to remember that changes in mass only occur because a gas is released into the atmosphere or because a gas from the atmosphere is used as a reactant, the atoms are not created or destroyed. , just reorganized.
What is a mass comparison?
The mass balance equation simply states that total mass is always preserved in any system. That is, the total input mass = the total output mass + the total accumulated mass in the system. Mass balancing can be performed for various components in the input and output streams.
What is the law of conservation of mass in simple terms?
The law of conservation of mass states that no mass can be created or destroyed by a chemical reaction. The amount of material cannot therefore be changed.
What does the law on the conservation of matter say?
The law of conservation of matter states that in any system closed to the transfer of matter, the amount of matter in the system remains constant. Answer b The law of conservation of matter states that in chemical reactions the total mass of the products must be equal to the total mass of the reactants.
Does a balanced chemical equation satisfy the law of conservation of mass?
Every chemical equation satisfies the law of conservation of mass, which states that matter can neither be created nor destroyed. Use the product and reactant coefficients to equalize the number of atoms in an element on either side of a chemical equation.
What is the law of multiple proportions?
Examples of the law of multiple ratios The ratio of the masses of oxygen combined to the solid mass of hydrogen in H2O and H2O2 shows an integer ratio. The ratio of the masses of oxygen combined to the solid mass of copper in CuO and CuO2 shows an integer.
What is the law of certain proportions?
In chemistry, the law of specific proportions, sometimes Proust's law or the law of specific composition or the law of constant composition, states that a given chemical compound always contains its constituents in a fixed ratio (mass) and does not depend from the Origin and method of preparation.
What is the example for the crowd?
Examples of mass. For example, a person or object may be weightless due to the lack of gravity on the moon, but the same person or object maintains the same mass regardless of its position. Knowledge of the masses is vital in science.
Which Equation Shows A Conservation Of Mass
![Law of Conservation of Mass - Fundamental Chemical Laws, Chemistry]()
 Daniel Johnston
Daniel Johnston 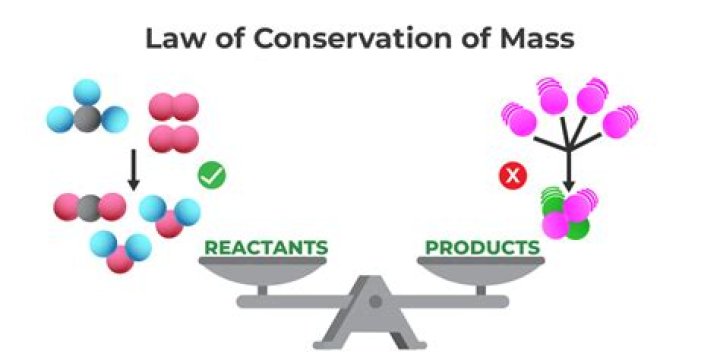
 Daniel Johnston
Daniel Johnston 