Specific Heat Of Brass - How To Discuss
 Daniel Johnston
Daniel Johnston 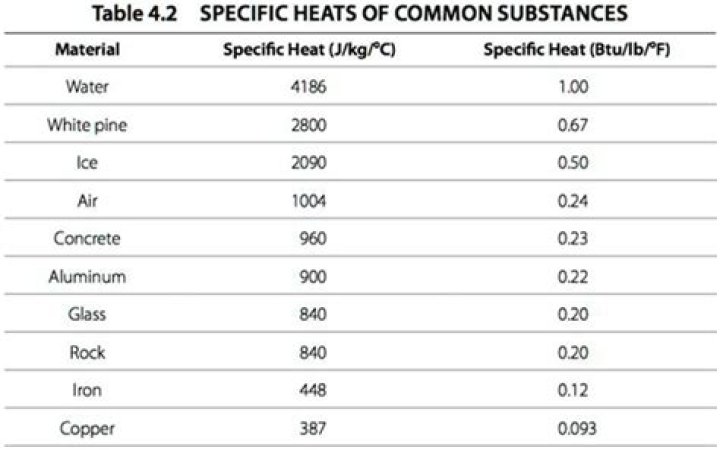
Specific Heat Of Brass
What is the specific heat of brass?
| Specific heat and molar heat capacity for various substances at 20 ° C.
Powder c i J / gm K c i cal / gm K or Btu / lb F| Buyers | 0.386 | 0.0923 |
| Brass | 0.380 | 0.092 |
| gold | 0.126 | 0.0301 |
| tube | 0.128 | 0. ### 0305 |
| Brass | 920 | 0.220 |
| calculation | 841 | 0.201 |
| Loose | 554 | 0.122 |
| Clay | 878 | 0. ### 210 |
What is the highest specific heat capacity?
Water has the highest specific heat capacity of all liquids. Specific heat is defined as the amount of heat that a gram of a substance must absorb or give up in order to change its temperature by one degree Celsius. In the case of water, this amount is one calorie, or 4,184 joules.
What is the formula for specific heat?
Specific heat is the amount of heat needed to increase one gram of a substance by one degree Celsius or Kelvin. The formula for specific heat is the amount of heat absorbed or emitted = mass x specific heat x temperature change.
Which metal has the highest specific heat capacity?
Which metal heats up the fastest, aluminum, copper or silver?
What is the specific heat capacity?
i?
The SI units are the specific heat capacity (symbol: c) the amount of heat in joules needed to increase 1 gram of a substance by 1 Kelvin. It can also be expressed in J / kg K. The specific heat capacity can also be expressed in units of calories per gram Celsius.
What is the specific heat of carbon?
0.71 J / g
Which material has the highest specific heat?
Gray Granite With a specific heat nearly three-tenths of that of water, gray granite has by far the highest specific heat in the material. This means that it has a large heat capacity and requires a relatively large amount of heat energy to raise the temperature.
Which solid has the highest specific heat?
I don’t know of anything that has a higher specific heat than water (although I don’t want to be dogmatic about water being the highest). For a very specific heat resistant fabric, look for something like ice or some other fabric that is durable and has a high water content at the desired temperature.
Can the heat capacity be negative?
If the system loses energy, for example by radiating energy into space, the average kinetic energy increases. Thus, if a temperature is defined by the average kinetic energy, the system can be said to have a negative heat capacity.
What is the specific heat of graphite?
Thermodynamics: heat and enthalpy
What is the specific heat of ice?
The specific heat capacity or amount of heat required to raise the temperature of a certain substance in a certain form by one degree Celsius is 4.187 kJ / kgK for water, 2.108 kJ / kgK for ice and 1.996 for water vapor (steam) kJ / kgK.
What is the specific heat capacity of copper?
The heat capacity of copper Cp (298.15 K) is 24,438 J / molK.
What is the specific heat of silver?
Answer and explanation: The specific heat of silver is 0.233 J / gK. This can also be written with 2.46 J / g degrees Celsius. This means 0.233 J of heat is needed for
What is the lowest specific heat capacity?
Specific heat capacity of the substance at 25oC in J / gC| to see | 0.21 |
| tube | 0.160 |
| mercury | 0.14 |
| gold | 0.129 |