Ses2 Lewis Structure - How To Discuss
 Amelia Brooks
Amelia Brooks 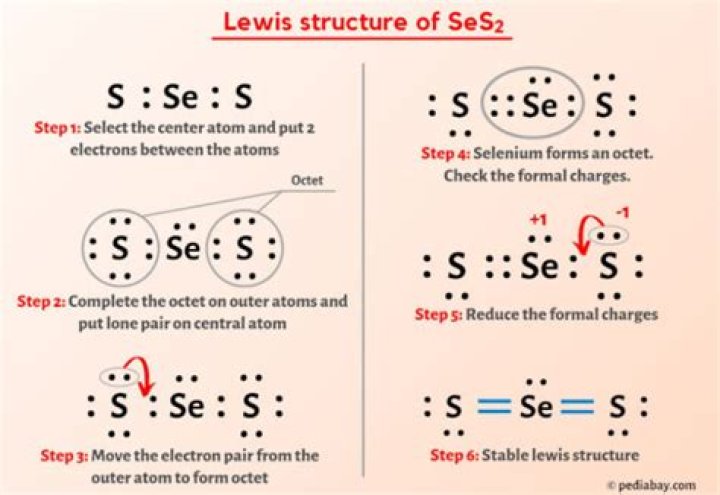
Ses2 Lewis Structure
How many valence electrons must be in the Lewis structure of SeS2? 3
How many valence electrons can be formed in the Lewis structure of SeS2?
Thanks!
SSe = S with 3 isolated pairs in the first S, 1 isolated pair in the Se and 2 isolated pairs in the second S
Carbon has 4 valence electrons because it belongs to group IV. Hydrogen has 1 valence electron. To make it, each atom must have an all-byte structure. For carbon, it must have a total of 8 valence electrons. For hydrogen, it must have a total of 2 valence electrons. One is formed when carbon and hydrogen divide electrons to form 4 covalent bonds.