Secl4 Lewis Structure - How To Discuss
 Jessica Cortez
Jessica Cortez 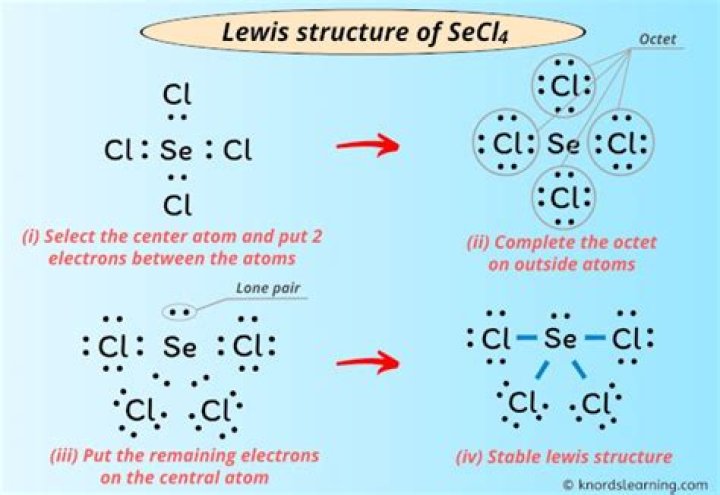
Secl4 Lewis Structure
Need help with chem? 3
SeCl4, SeF6
What is the electron group geometry of each molecule, what is the molecular geometry of each molecule, and is the molecule polar or non-polar?
Lever
Actonal
First you need to calculate the total number of valence electrons (ves) for the molecule.
SeCl4 ...
Se x 6 times 1 atom = 6 times (6 times, because Se is part of group 6)
Atom 4 Cl x 7 times = 28 times
Add time ... Total 28 + 6 = 34 times
To draw the Lewis structure:
I'm not sure what you learned, but I found it easy to draw a simple link.
It would be in the middle with 4 chlorine around the Se atom, so it would look like this:
Tomorrow
|
ClSeCl
|
Tomorrow
Now each bond consists of 2e, so far there are 4 bonds or 8 electrons.
Remember that each element must be accompanied by an eighth note and so far the only element if
Cl atoms must also have 8, but so far they have only 2 between the bonds they form to complete their bytes. Cl atoms need to be surrounded by single pairs, in particular, they must have 3 single pairs to give you a total of 8 electrons (2 bonds ... 2 out of each pair).
So now each is a sixth of Cl.
(The reason I say 6 and 8 is because the electrons are already in a bond, in other words if you count the electrons it won't count twice)
... and 4 Cl, so ... 6x4 = 24
He also has an eighth, so tomorrow
24 + 8 = 32 ... but the total should be 34, so where did the other 2 go?
They were placed as a couple in the central part of Se's housing development.
Now, to get electron group geometry, count the number of bonds and the number of solitary pairs in the central atom.
4 links + 1 isolated pair = 5 electronic domains Geometry is a triangular pyramid according to 5 electronic domains.
This only happens with 5 bonds, but since there are 4 bonds and only one pair, the geometry of the molecule will be lead. Due to its shape, SeCl4 is a polar molecule.
If you do SeF6.
If ... 6 out of 42
Total ve = 48 ve
If in the middle ... 6 F surrounds it and 3 isolated pairs in each F.
So here are a total of 12 .... 2 each link from F.
And F has a total of 36º ..... each F has 6 isolated pairs.
36 + 12 = 48
The SeF6 will have an electronic octahedron and molecular geometry.
This page can help you.
D:
Need help with chem?
SeCl4, SeF6
What is the electron group geometry of each molecule, what is the molecular geometry of each molecule, and is the molecule polar or non-polar?
Secl4 Lewis Structure
Secl4 Lewis Structure
Lewis structure of Secl4
Yes, it is correct.