Sbf5 Polar Or Nonpolar - How To Discuss
 Rachel Newton
Rachel Newton 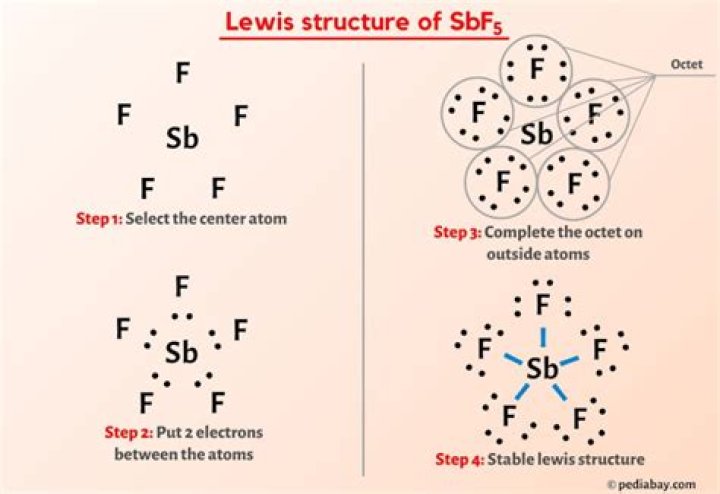
Sbf5 Polar Or Nonpolar
Which of the following molecules is polar? 3
A) CH4
B) SbF5
C) CO2
D) AsH3
Can you easily explain that you accept your answer because I never believed that chemistry is like that?
Thank you very much.
updateThe answer key states that the correct answer is D. I don't know what to do with it.
I think this is really your Ash3. SbF5 forms the structure of a pyramid, which means that in e the polar bonds are evenly distributed between antimony and hydrogen, so the net polarity of the molecule is z.
ASH3, on the other hand, is a pyramid-shaped molecule with Nic at the top and three hydrogen atom heads pointing, with a pair of electrons pointing to the fourth corner of the tetrahedron with Nic in the middle. ۔ All ASH bonds have a polarity in which the positive end is at H and the negative end is at As, and the pair of electrons in the fourth corner make it more polar.
Sbf5 Polar Or Nonpolar
Sbf5 Polar Or Nonpolar
AsH3 is ■■■■■■■■ and its molecular form is similar to ammonia (NH3) because they belong to the same group. These two molecules are polar because nitrogen and nickel atoms have only one pair of electrons, which is the source of negative charge. The opposite part of the molecule has a positive charge.
But what helps?
Marco
CH "is a tetrahedron, apolar
SbFÃ ... is a triangular pyramid, not a pole.
COA is linear, not polar.
AsHƒ is a triangular pyramid with a base of one ace and three Hs, polar
If you've never taken a chemistry class, I'm not sure you should know this.
B.
Pole means equal on all sides. If you haven't taken a chemistry class yet, it can be hard to explain. When you draw the formula, you will notice that B is not compatible.