Oxygen Ion Charge - How To Discuss
 Isabella Browning
Isabella Browning 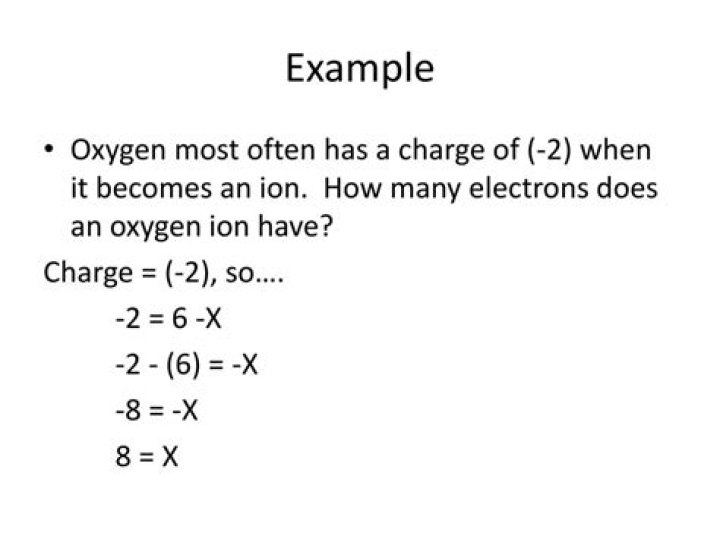
Oxygen Ion Charge
What is the charge of an oxygen ion with 9 electrons and 8 protons?
Cost of 2 (10 + 8 = 2).
In this perspective, what is the charge of an ion with 8 protons and 10 electrons?
Explanation: With eight protons we are necessarily looking at oxygen, but with 10 electrons the net charge of the ion is minus two. The closest thing to writing the symbol in this answer is O2− (the character must be part of the title).
What is the net charge of a proton ion 9?
The element fluorine has 9 protons and 9 electrons. In the figure on the right it has one electron and now it has 10. Since it has 10 negative particles but only 9 positive, its net charge is -1.
Second, which ion has 8 protons, 9 neutrons and 10 electrons?
Oxygen ionsHow many electrons do oxygen ions have?
Oxygen has the atomic number eight. This means that the nuclei of all oxygen atoms have eight protons. A neutral oxygen atom also has eight electrons. In a 2-charge oxygen ion, the number of protons is still 8, but the number of electrons is 10.
Is oxygen an ion?
Oxygen, O. Oxygen belongs to group 6. It has six electrons in its outer shell. In reactions it receives two electrons from one or two other atoms and forms an oxidation, O 2.
How do we find electrons?
The number of protons, neutrons and electrons in an atom can be determined according to simple rules. The number of protons in the atomic nucleus equals the atomic number (Z). The number of electrons in a neutral atom equals the number of protons.
Is oxygen a cation or an anion?
Oxygen itself is neutral (unloaded). To be considered a cation or anion, there must be a cost associated with it. Although it really depends on whether the anode / cathode is seen as positive or negative (both can be both even though the other should be the opposite).
Which element has a charge of 2 when it has 10 electrons?
Table of current elementary charges Number of elementary charges 9 Fluorine 1 10 Neon 0 11 Sodium 1+ 12 Magnesium 2+
How high is the total oxygen charge?
The ionic charge of oxygen is 2. You can see this because oxygen is in group 6, so it has 6 valence electrons. Since with a full outer shell of 8 electrons it takes 2 more electrons to become neutral, its charge is 2 (since adding electrons results in a negative charge).
Is oxygen neutral or ions?
A neutral oxygen atom is represented by O. It has atomic number 8. Therefore, it contains 8 electrons and 8 protons. When oxygen forms an ion, it accepts 2 electrons to become an O2 ion.
What is ionic notation?
If you enter an Ionic symbol, the symbol is first written with one or two letters, followed by a title. The head has the number of charges on the ion followed by a + (for positive ions or cations) or (for negative ions or anions). Neutral atoms have zero charge, so no direction is given.
Which element has 7 protons, 8 neutrons and 10 electrons?
A nitride has 7 protons, 8 neutrons and 10 electrons.
What is the total charge of 8 protons, 10 neutrons and 8 electrons?
That is, an oxygen atom usually has 8 protons and 8 electrons, this ion has 10 electrons (10 charges) and 8 protons (+8 charges), which gives it a charge of 2 (10 + 8 = 2).
What is the symbol of the atom with 9 protons, 10 neutrons and 9 electrons?
If you look at the periodic table you can see that fluorine has 9 protons. Since the number of electrons equals the number of protons, fluorine also has 9 electrons. Meanwhile, you emit 19 masses, minus 10 neutrons, you emit 9 protons or electrons. So the atom would be fluorine.
Which ion has 9 protons, 9 neutrons and 10 electrons?
Fluorine
Which element has 8 protons, 8 electrons and 11 neutrons?
Oxygen
What is the formula of the nitride ion?
Final results for the nitride ion formula. The formula of the nitride ion is N3a. The term nitride ion refers to any chemical compound that contains nitrogen ions, which have a negative wood oxidation state.
Which element has the same number of energy levels as nitrogen?
Electrons Element of atomic number Energy levels or shells 7 Nitrogen (N) 2 8 Oxygen (O) 2 9 Fluorine (F) 2 10 Neon (Ne) 2