Osmosis Can Be Defined As
 Daniel Johnston
Daniel Johnston 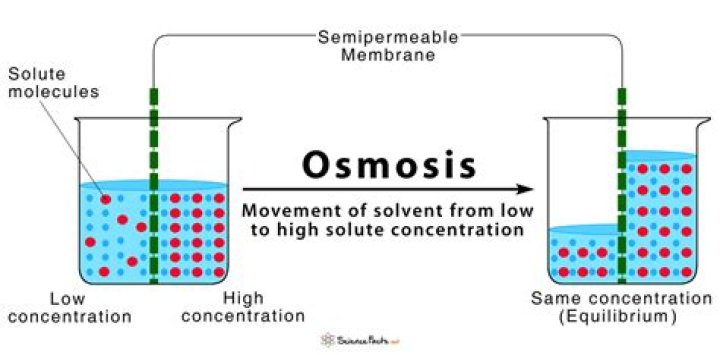
Osmosis can be defined as a process in which water is transported over a semipermeable membrane from a solution of high concentration to a solution of low concentration. Water or other solvents can travel through a semipermeable barrier on their own.
![:eight_pointed_black_star: :eight_pointed_black_star:]() Osmosis Definition
Osmosis Definition
Osmosis is defined in biology as the net flow of water molecules through a semipermeable membrane (e.g., a cell membrane) from a region with a greater water potential to a region with a lower water potential. Additional definitions of osmosis include the following:
The process by which a solvent diffuses through a semipermeable membrane from a low-concentration area to a high-concentration area.
This is how easy it is for water to move from a hypotonic solution to a hypertonic solution through a semipermeable shield.
Osmosis is defined similarly in chemistry. It is the transit of a pure solvent from a solution with a lower concentration of solutes to one with a higher concentration of solutes when the two solutions are separated by a membrane that blocks the passage of solute molecules but enables the passage of solvent molecules.
When a solution is isolated from a pure solvent by a membrane that allows only the solvent through but not the solute, the solution tends to dilute. This process can be halted by increasing the pressure in the solution by a predetermined amount, referred to as the osmotic pressure.
In 1886, the Dutch-born chemist Jacobus Henricus van 't Hoff demonstrated that when a solute is dilute enough that its partial vapor pressure above the solution obeys Henry’s law (i.e., is proportional to its concentration in the solution), osmotic pressure varies with concentration and temperature in the same way that a gas does.
Summary
This relationship resulted in formulae for calculating the molecular weights of solutes in dilute solutions based on the solvent’s freezing point, boiling point, or vapour pressure.
![:eight_pointed_black_star: :eight_pointed_black_star:]() What exactly is osmosis?
What exactly is osmosis?
When we divide a dilute solution from a pure solvent of the same kind using a membrane, we find that the solvent molecules move from the dilute solution to the concentrated solution. This motion is repeated until balance is attained. Osmosis is the mechanism through which solvent molecules flow owing to a change in concentration.
By adding external pressure on the membrane, we may halt the passage of solvent molecules across it. As mentioned previously, the osmotic pressure is the pressure that simply stops the flow of solvent particles.
True, osmotic pressure is a colligative property, as it is proportional to the solution’s concentration. In other words, osmotic pressure is the additional pressure required to prevent osmosis.
Experimental evidence indicates that this pressure is exactly proportional to the solution’s molarity (number of mole’s solute per liter of solution) at a certain temperature. Isotonic solutions are two liquids that have the same osmotic pressures at a given temperature. Learn more about the many types of solutions available here.
![:small_red_triangle_down: :small_red_triangle_down:]() Proof Mathematical
Proof Mathematical
π = CRT
C= Denotes the solution’s molarity.
R=Universal Constant of gas:
T=Temperature
π=osmotic pressure
The molarity of a solution is the mole fraction of solute per litre. If w2 is the mass of a solute of molar mass M2 in a volume V solution, we may write,
π=M2V/w2RT
We frequently use the following equation to determine the molar mass of complex molecules such as proteins, polymers, and other macromolecules. Scientists choose to utilize this for biomolecules since it exhibits a considerable value at ambient temperature.
![:small_red_triangle_down: :small_red_triangle_down:]() Is osmosis a possible cause of cholera?
Is osmosis a possible cause of cholera?
Ans: Osmosis also enables bad things to occur. Without osmosis, cholera would not be conceivable. The choleric bacteria colonize our intestines and begin to invert the ionic orientation of the intestinal cells. In other words, it alters the manner in which ions and, consequently, water are transported through our intestines. This indicates that cholera pulls off a flawless coup.
Second, this accelerates the pace of dehydration. Not only are you unable to absorb water, but you are being practically emptied dry. This is why cholera may be fatal so rapidly, as it is not dependent on the amount of water consumed.
Summary
To begin, when our ions’ orientations are reversed, the intestinal cells lose their ability to absorb water. Now, osmosis occurs in the other direction, with water moving out of our intestinal cells and into them. This is what produces the infamously lethal watery diarrhea associated with cholera.
![:eight_pointed_black_star: :eight_pointed_black_star:]() Osmosis Mechanism
Osmosis Mechanism
Osmosis is a type of diffusion that is distinct from others. Water, like other chemicals, goes from a concentrated region to a depleted area. The obvious issue is what causes water to flow in the first place.
Consider a beaker with two sides or halves separated by a semipermeable membrane. The water level is identical on both sides of the membrane, but the concentration of a dissolved material, or solute, is different and cannot spike the barrier.
Water is present in varying proportions on both sides of the membrane if the volume of solution on either side of the membrane is the same but the solute concentrations differ. If there is a greater concentration of solute in one location, there must be less water; if there is a lesser concentration of solute in one place, there should be more water.
Consider two full cups of water as an illustration. One includes a single spoon of sugar, while the other holds a quarter cup. Sugar takes up a lot more room in the second cup, thus the first cup has a lot more water in it since there is so much sugar in it.
Recall that the beaker contains a mixture of solutes on both sides of the membrane. Diffusion is based on the premise that molecules move and will spread equally across the medium if they can.
Like a result, crossing the membrane to the less concentrated side, water diffuses along its concentration gradient. Osmosis—the process by which water diffuses across a membrane—will continue until the concentration gradient of water equals zero or until the hydrostatic pressure of the water equals the osmotic pressure. This means that in the beaker example, the level of fluid in the side with a greater solute concentration will increase.
| No | Key Points |
|---|---|
| 1 | Osmosis occurs as a result of the gradient of water concentration across the membrane, which is inversely proportional to the concentration of solutes. |
| 2 | Osmosis happens until the concentration gradient of water equals zero or the hydrostatic pressure of water equals the osmotic pressure. |
| 3 | Osmosis occurs when a solution has a concentration gradient of a solute but the membrane prevents the solute from diffusing. |
| Key Terms | |
|---|---|
| Solvent: | Any material dissolved in a liquid solvent to form a solution |
| Osmosis: | The net movement of solvent molecules from a region with a high solvent potential to a region with a low solvent potential via a partially permeable membrane |
| Semipermeable membrane: | A type of biological membrane that allows certain molecules or ions to pass through it via diffusion and, on occasion, specialized facilitated diffusion |
Summary
However, only substances capable of diffusing past the membrane will do so. The solute cannot diffuse across the barrier in this case, but the water can. In this system, water exhibits a concentration gradient.
![:eight_pointed_black_star: :eight_pointed_black_star:]() Osmotic types of solutions
Osmotic types of solutions
A solution is fundamentally composed of two components: solute (stuff to be dissolved) and solvent (the component that dissolves the solutes). The concentrations of the elements of two solutions shall be used to assess whether one solution is isotonic, hypotonic, or hypertonic in comparison to another.
![:small_red_triangle_down: :small_red_triangle_down:]() Isotonic
Isotonic
An isotonic solution is one in which the concentration of solutes is almost identical to that of another solution. For example, an isotonic cell to the external solution indicates that both the internal and extracellular fluids have the same osmotic pressure and water potential. There will be no net flow of water molecules between the cell and the surrounding fluid in this situation.
![:small_red_triangle_down: :small_red_triangle_down:]() Hypertonic
Hypertonic
A hypertonic solution is the opposite of a hypotonic solution. A hypertonic solution will have a higher concentration of solutes and a lower concentration of water than the other solution. When a cell is submerged in a hypertonic solution, water will escape to dilute the solution outside the cell.
![:small_red_triangle_down: :small_red_triangle_down:]() Hypotonic
Hypotonic
A hypotonic solution is one that has a lower osmotic pressure (or contains fewer solutes) than another solution. In this situation, water flows toward areas with a lower concentration of water or toward areas with a higher concentration of water in order to dilute the solution.
Water will cross the cell membrane into the body’s more pure solvent if the fluid around it is hypotonic, for example.
![:eight_pointed_black_star: :eight_pointed_black_star:]() Water Osmosis and Net Movement
Water Osmosis and Net Movement
Osmosis is the net flow of water across a selectively permeable membrane caused by a difference in the concentrations of solutes on the membrane’s two sides. A membrane that is selectively permeable enables unfettered passage of water but not of solute molecules or ions.
Different solute molecule concentrations result in varying quantities of free water molecules on each side of the membrane. On the side of the membrane with a higher concentration of free water (i.e., a lower concentration of solute), more water molecules will pass through the pores in a given time interval.
The critical aspect of osmosis to remember is that water flows from a solution with a lower solute concentration to a solution with a greater solute concentration. This indicates that water flows through a membrane in response to changes in molarity.
Osmosis is unaffected by the size of the solute particles. Equilibrium is established when sufficient water has passed over the membrane to equalize the solute content on both sides, at which time net water flow ceases. Here is a basic illustration of these principles:
Two containers of identical volume are separated by a membrane that enables water to move freely but completely prevents solute molecules from passing. Assume Solution A contains three albumin (MW 66,000) molecules and Solution B contains fifteen glucose molecules.
Summary
More strikes results in more molecules moving through the pores, resulting in net water diffusion from the compartment with the highest concentration of free water to the compartment with the lowest concentration of free water.
![:eight_pointed_black_star: :eight_pointed_black_star:]() Osmosis Types
Osmosis Types
Before we discuss the distinction between Endosmosis and Exosmosis, let’s review the definition of “osmosis.” Osmosis is the process by which a solvent (such as water) moves from a solution with a low solute concentration to one with a greater solute concentration across a semipermeable membrane. When equalization occurs on both sides of the membrane, the flow of this solution ceases.
There are two forms of osmosis: exosmosis and endosmosis.
![:small_red_triangle_down: :small_red_triangle_down:]() Exosmosis
Exosmosis
When a cell is put in a hypertonic solution, the water inside the cell begins to evaporate, and the cell becomes flaccid. Exosmosis is the process through which water leaves the cell. This occurs because the concentration of solutes in the surrounding solution is greater than the concentration within the cytoplasm.
![:small_red_triangle_down: :small_red_triangle_down:]() Endosmosis
Endosmosis
When a cell is immersed in a hypotonic solution, the water within the cell expands. Endosmosis is the term used to describe the flow of water within a cell. This occurs because the outer solution contains a lower concentration of solutes than the cytoplasmic solution.
![:eight_pointed_black_star: :eight_pointed_black_star:]() Osmosis examples
Osmosis examples
Osmosis is a type of diffusion in which only water molecules pass through a semi-permeable membrane from an area of high concentration to a region of low concentration.
![:small_red_triangle_down: :small_red_triangle_down:]() Osmosis in animal cells
Osmosis in animal cells
Osmosis is critical in biological systems because many biological membranes are semipermeable, resulting in a variety of physiological consequences.
For instance, when animal cells are subjected to a hypertonic environment (with a decreased water content), the water is drained from the cells, resulting in cell shrinkage. Crenation is the term used to describe this condition.
In contrast, when animal cells are put in a hypotonic environment (or one with a greater water content), water molecules enter the cells, causing them to swell. If osmosis is allowed to proceed unabated, the cells will finally explode.
![:small_red_triangle_down: :small_red_triangle_down:]() Osmosis in plant cells
Osmosis in plant cells
Plant cells do not rupture as a result of excessive water inflow. Through their cell walls and vacuoles, plants are able to counteract excessive osmosis. The cell wall provides osmotic pressure on the plant cell, therefore stabilizing it. Indeed, it is osmotic pressure that causes plants to remain plumb.
Additionally, the big vacuole within the plant cell aids in osmoregulation, a regulatory process in which water potential is adjusted to maintain an appropriate osmotic pressure within the cell.
Summary
However, water outflow does not protect plant cells. In a hypertonic environment, a plant’s cell wall cannot prevent water loss. This results in the cell contracting or becoming flaccid.
![:eight_pointed_black_star: :eight_pointed_black_star:]() Osmotic and Hydrostatic Pressure Calculation
Osmotic and Hydrostatic Pressure Calculation
Osmosis, the circulation of water across a membrane in reaction to the concentration of solute molecules on either side, creates an osmotic pressure throughout the membrane. Osmotic pressure is defined as the hydrostatic pressure necessary to stop the flow of water.
Consequently, osmotic and hydrostatic pressures are comparable in all practical terms. A membrane might be a synthetic lipid bilayer, a plasma membrane, or a layer of cells.
The following equation approximates the osmotic pressure P of a dilute solution:
P equals RT (C1 + C2 +… + Cn).
where R signifies the gas constant, T denotes the absolute temperature, and C1. Cn signify the solutes’ molar concentrations (ions and molecules).
In a similar fashion, the osmotic pressure across a membrane separating two fluids is as follows:
P equals RT (C)
Where C denotes the solute concentration difference between the two solutions. Thus, if the membrane is permeable to water but not to solutes, the osmotic pressure is proportional to the solute concentration differential across the membrane (the proportionality factor is RT).
![:eight_pointed_black_star: :eight_pointed_black_star:]() How is Osmotic Pressure Defined?
How is Osmotic Pressure Defined?
The final colligative feature of solutions that we shall discuss is critical for biological systems. It is accomplished by osmosis, the mechanism by which solvent molecules flow through particular membranes but solute particles do not.
When two solutions of varying concentrations are present on each side of these membranes (referred to as semipermeable membranes), solvent molecules have a tendency to migrate from the more dilute solution to the more concentrated solution until their concentrations are equal.
This is referred to as osmotic pressure. External pressure can be applied to a solution to cease the flow of solvent; the pressure required to halt osmosis is equal to the solution’s osmotic pressure.
Osmolarity (osmol) is a unit of measurement used to express the total number of particles in a solution in order to calculate the osmotic pressure. It is defined as a solute’s molarity multiplied by the number of particles formed when a formula unit of the solute dissolves (represented by I
osmol=M×i
When more than one solute is present in a solution, the separate osmolarities combine to form the solution’s total osmolarity. Equally osmolar solutions have the same osmotic pressure. When two solutions with varying osmolarities are placed on opposing sides of a semipermeable membrane, solvent will move from the lower-osmolarity solution to the higher-osmolarity solution.
Summary:
By applying counterpressure to the high-osmolarity solution, the solvent transfer will be slowed or halted. Reverse osmosis is the process of forcing solvent from a high-osmolarity solution into a low-osmolarity solution. Reverse osmosis is used to convert seawater to drinkable water in areas where fresh water supplies are rare.
Frequently Asked Questions - FAQs
People asked many questions about osmosis. We discussed a few of them below:
![:one: :one:]() What is the definition of a semipermeable membrane?
What is the definition of a semipermeable membrane?
Semipermeable membranes are biological membranes that allow some molecules or ions to flow through them.
![:two: :two:]() How is reverse osmosis defined?
How is reverse osmosis defined?
Reverse osmosis is a natural phenomena in which water moves in the opposite direction of normal osmosis. This sort of osmosis is used to remove the bulk of pollutants from water by forcing it through a semi-permeable membrane under pressure.
![:three: :three:]() What is osmosis and what is class 12 osmotic pressure?
What is osmosis and what is class 12 osmotic pressure?
When a semi-permeable membrane is sandwiched between a solvent and a solution, the solvent molecules pass across the membrane from the pure solvent to the solution. Osmotic Pressure: By providing additional pressure to the solvent side, the flow of solvent from the solvent side to the solution side (through a semi-permeable membrane) can be prevented.
![:four: :four:]() What is the term “osmotic pressure”?
What is the term “osmotic pressure”?
The osmotic pressure is defined as the lowest pressure that must be given to a solution in order to prevent solvent molecules from flowing through a semipermeable barrier. The osmotic pressure of a solution is proportional to the solute particle’s molar concentration.
![:five: :five:]() In biology, what is osmosis?
In biology, what is osmosis?
Osmosis is the process by which a solvent is transported via a semipermeable membrane that separates two solutions with varying concentrations of solutes. Osmosis is the process by which a solvent travels from a solution with a low solute concentration to a solution with a high solute concentration.
![:six: :six:]() What distinguishes osmosis from diffusion?
What distinguishes osmosis from diffusion?
Osmosis is the process by which solvents migrate from an area of low solute concentration to a region of high solute concentration across a semi-permeable membrane. On the contrary, diffusion occurs without the presence of a semi-permeable barrier, and molecules migrate from a location of greater concentration to a region of lower concentration.
![:seven: :seven:]() Are dull cells capable of osmosis?
Are dull cells capable of osmosis?
Yes, dull cells display osmosis as well. When a dull cell is submerged in a hypotonic solution, water enters the cell, causing it to swell.
![:eight: :eight:]() What is osmosis’ primary function?
What is osmosis’ primary function?
Osmosis contributes to the stability of the organism’s internal environment by balancing the amounts of water and intracellular fluids. Additionally, nutrients and minerals enter the cell via osmosis, which is required for cell viability.
![:nine: :nine:]() What does osmosis require?
What does osmosis require?
Accordingly, for assimilation to happen, the layer should be penetrable to water however impermeable to the solute, and the solute fixation on the different sides of the film should be unique.
![:keycap_ten: :keycap_ten:]() What is the term “forward osmosis”?
What is the term “forward osmosis”?
Forward osmosis is a natural process that occurs on a regular basis in our environment. It is a form of osmosis in which water is separated from dissolved solutes using a semi-permeable membrane. This sort of osmosis is frequently employed in wastewater treatment and osmotic energy generation, among other applications.
Conclusion:
The net flow of solvent through a semipermeable is known to as osmosis. It is analogous to diffusion in that the movement is downward, from a higher to a lower concentration. Nonetheless, in assimilation, the development should occur over a semipermeable boundary. Without this component, osmosis cannot be termed such.
While diffusion is frequently illustrated as the net movement of solvents between two solutions, osmosis is concerned with the net movement of solvent molecules, such as water molecules. The difference in water molecule concentration between the two sides of the membrane is what causes the water to migrate in order to equalize the two regions’ concentrations.