O3 Lewis Structure - How To Discuss
 Amelia Brooks
Amelia Brooks 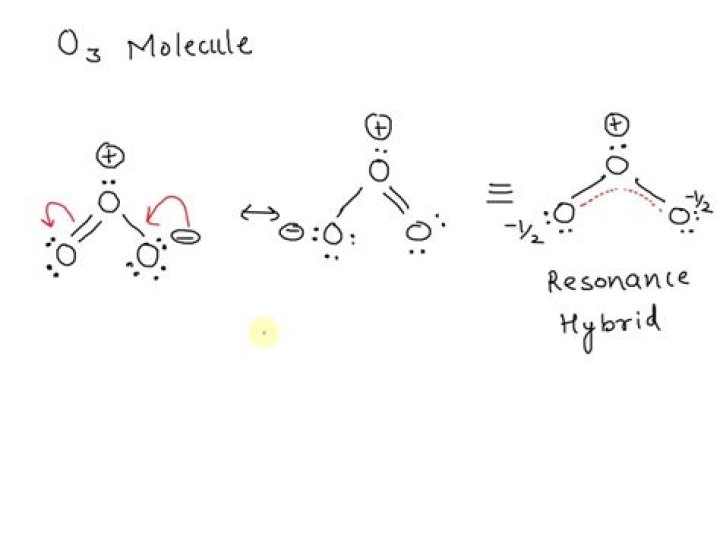
O3 Lewis Structure
What is the Lewis dot structure of O3? 3
Counting all the electrons, it is difficult to say whether you mean O3 or O-3.
I don't think that's really going to happen. An oxygen atom does not have three electrons. On Sabellol 2, there is only room for two more electrons. Adding two electrons gives the total energy level. This is the number of electrons in the oxygen atom. Three O 2 for one oxide ion.
Ozone has double bond and single bond in the Lewis structure. In fact, it has two identical bonds, each with a bonding order of 1.5 due to delicate pie bonds formed from a non-hybrid portal on top of each oxygen atom.
Adding another electron to get O3 get will get one weird electron and one. The Lewis structure is said to have single bonds between oxygen atoms. The terminal atom will have one byte of electrons and the central atom will have single pairs and single electrons. It will not be a stable molecule.
There I reiterate that neither O3 nor O-3 is possible.
Lewis structure for O3.
Lewis structure of O3.
This page can help you.
D:
What is the Lewis dot structure of O3?
O3 Lewis Structure
O3 Lewis Structure
S is counting all the electrons. It is difficult to say whether you mean O3 or O 3. Is the charge of ozone negative or is the charge of oxygen atom negative in three?
I don't think that's really going to happen. An oxygen atom does not have three electrons. At sub-level 2, there is room for only two more electrons. The addition of two electrons results in a total energy level. This is the number of electrons in the oxygen atom. Three O 2 for one oxide ion.
Ozone has a double bond and a single bond in the Lewis structure. In fact, it has two identical bonds, each with a bonding order of 1.5 due to de-localized pie bonds formed by non-hybrid portals on top of each oxygen atom.
Adding another electron to get O3 ملے gives the odd number of electrons and one. The Lewis structure is said to be the only bond between oxygen atoms. The terminal atom will have one byte of electrons and the central atom will have a single pair and a single electron. It will not be a stable molecule.
There I repeat that neither O3 O nor O 3 is possible.