No3cl Lewis Dot Structure - How To Discuss
 Andrew Campbell
Andrew Campbell 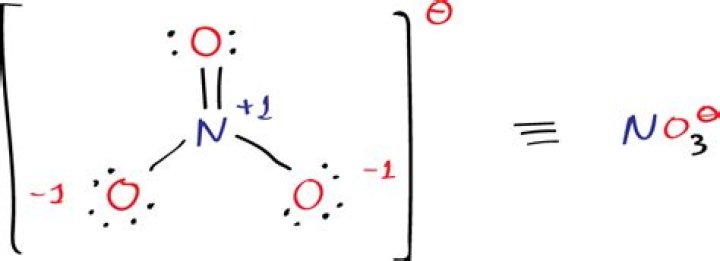
No3cl Lewis Dot Structure
Lewis structure of NO3Cl and regular charge of each molecule? ۔
I need to know the Lewis dot structure of NO3Cl ????
For travel, you do not have a formal charge on the molecule, but a formal charge on every element of the molecule.
NO3Cl can be combined with chlorine nitrate and has the following composition.
Clone = O
........ |
....... or
There are three isolated pairs of chlorine. The oxygen atom of a clone has two separate pairs. Oxygen N = O has two wandering pairs, while the rest of the oxygen atoms have three wandering pairs. Nitrogen has no separate partner.
There are two resonant structures in which a double bond runs between two terminal oxygen atoms.
Official rate
Cl = 0.
O (ClON) = 0.
N = +1.
O (N = O) = 0.
Or (no) = 1.
FC = VE NBE 1 / 2BE .... where VE = valence electron, NBE = inbound electron, BE = bond electron
Lewis Structure No. 3cl
This page can help you.
D:
Lewis structure of NO3Cl and regular charge of each molecule?
I need to know the Lewis dot structure of NO3Cl ????
The answer is B0 and the answer is C1. It's straightforward, so am I. That's the question I asked myself. I'm going to LSU. If I need more help send me an email. Well someone else's answer was really bad for me for a second !!!!!!
No3cl Lewis Dot Structure
No3cl Lewis Dot Structure
Lewis structure of NO3Cl and regular charge of each molecule? 3
Do I need to know the Lewis dot structure for NO3Cl ????
For this, you have no formal charge on the molecule, you have a formal charge on every element of the molecule.
NO3Cl can be mixed with chlorine nitrate and has the following composition.
Clone = O
........ |
....... or
Chlorine has three isolated pairs. The oxygen atom of ClON consists of two isolated pairs. N = O Oxygen has two wandering pairs, while the rest of the oxygen atoms have three wandering pairs. Nitrogen has no isolated partner.
There are two resonant structures in which a double bond moves between two terminal oxygen atoms.
Official rates
Cl = 0
O (ClON) = 0
N = +1
O (N = O) = 0
Or (no) = 1
FC = VE NBE 1 / 2BE .... where VE = valence electron, NBE = unbound electron, BE = bond electron
This page can help you.
D:
Lewis structure of NO3Cl and regular charge of each molecule?
Do I need to know the Lewis dot structure for NO3Cl ????
Answer B is 0 and answer C is 1. That's right- this is me. That's the question I asked myself. I'm going to LSU, email me if you need more help. Well one minute answer is too bad for me !!!!!!