Nicl2 Lewis Structure - How To Discuss
 Amelia Brooks
Amelia Brooks 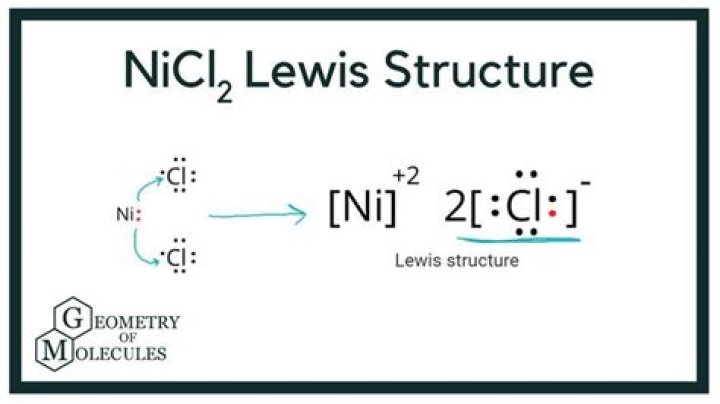
Nicl2 Lewis Structure
Would you like to make a Lewis NiCl2 scatter plot? 3
Nothing has 28 electrons, and my teacher told me that all the elements have the number of electrons in 2, 8, 8, 2 .... ss. When I follow this method and try to extract nickel, it has 8 external valence electrons. So should I mix 2 chlorine with nickel?
It is better to think of it as an ion, ie Ni 2 + plus two Cl ions. Ni 2 +, as you have guessed correctly, has a valence 8 e, but given that (Ni: [Ar] 3d 8), so you have a Ni 2 + ion There are around eight e s regular bytes.
Nicl2 Lewis Structure
Nicl2 Lewis Structure
If you draw a Lewis diagram of an ion like a sulfide ion, S = knowing that the sulfur atom has 6 electrons at the external energy level and of course the ion has 8 electrons which takes 2 of the other atoms, The structure therefore becomes Lewis' symbol S, which is surrounded by four pairs of electrons. Now, if you make a Lewis diagram for H2S, for example, every hydrogen, as you know, has one electron and Sulr has 6 electrons at the external energy level. And you probably know that the total charge of each molecule is z. So just make a sketch and don't worry about the formal charge, in that case H = S = H will be added and placed under the sulfur atom, and the shape of the molecule will be tilted 90 degrees.
Lewis structure of nickel 2
This page can help you.
D:
w Are you going to make a Lewis NiCl2 scatter plot?
Nothing has 28 electrons, and my teacher told me that all the elements have the number of electrons in 2, 8, 8, 2 .... ss. When I follow this method and try to extract nickel, it has 8 external valence electrons. So should I mix 2 chlorine with nickel?