Kmno4 Ionic Or Covalent - How To Discuss
 Daniel Cobb
Daniel Cobb 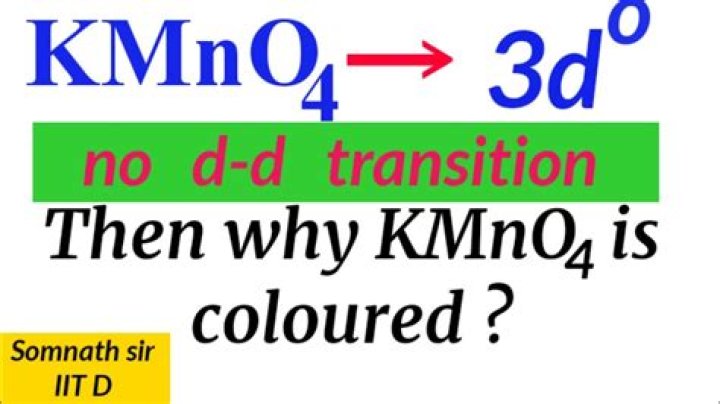
Kmno4 Ionic Or Covalent
Is KMnO4 ionic?
Potassium permanganate is an ionic compound devoid of potassium cation (K +) and permanganate anion (MnO4), in which the manganese atom is bonded to four oxygen atoms via a single bond and three double bonds.
Similarly, people ask: Is KMnO4 a pure compound?
Potassium permanganate is an inorganic compound, not a mixture and has the chemical formula KMnO4. It is associated with many names, the most common being potassium permanganate, mineral chameleon, state crystals, potassium chloride permanganate and hypermanganate.
Secondly, what kind of reagent is potassium permanganate?
Lesson Summary Potassium permanganate, also known as potassium permanganate or Condys crystals, is a chemical compound with the chemical formula KMnO4, consisting of a potassium ion (K +) and a permanganate ion (MnO4). It is a powerful oxidant, so it easily accepts or takes electrons from other substances.
Is KMnO4 also an oxidizing agent?
Potassium permanganate is an inorganic chemical compound with the formula KMnO4. It is a salt composed of K + and MnO4 ions. Formerly known as potassium permanganate or Condys crystals, it is a powerful oxidant. It dissolves in water to give intensely purple solutions.
What is KMnO4 used for?
Medical uses Potassium permanganate is used for a variety of skin conditions. These include fungal foot infections, impetigo, pemphigus, superficial wounds, dermatitis, and tropical wounds.
Why is KMnO4 a self-indicator?
So when all the permanganizations of the reaction are exhausted, the solution loses its pink color. This indicates the end of the reaction, and potassium permanganate is therefore called a self-indicator, since it is not only one of the reactants, but also an indicator.
Can i drink potassium permanganate?
Although not very toxic, concentrated solutions of potassium permanganate can be harmful to the skin. Permanganate is a strong oxidizing agent, similar to chlorine, so if it comes into direct contact with the skin it can cause irritation or burns. However, very little or no permanganate should enter the water that is filtered by a green sand system.
Is KMnO4 polar or non-polar?
Potassium permanganate is an ionic molecule. Polar or non-polar generally refers to covalent molecules. Potassium donated its electron to make polyatomic ion permanganate, so it’s best to ask. Potassium would be K +.
Why is KMnO4 colored?
Potassium permanganate (KMnO4) is colored because it absorbs light in the visible part of the electromagnetic spectrum. The source of the color is permanganation, as a metal charge transfer of the binder occurs between the oxygen orbitals and the empty orbitals on the metal.
What is acid or basic KMnO4?
What happens if potassium permanganate is added to the water?
When a solid sample of potassium permanganate is placed in water, the water molecules interact with the potassium cations and permanganate anions in the crystal to break the ionic bonds that hold the crystal together. Finally, a homogeneous solution of potassium permanganate in water is obtained.
How is KMnO4 processed?
Potassium permanganate (KMnO4) consists of pyrolusite ore (MnO2). The finely pulverized pyrolusite ore (MnO2) is fused with an alkaline hydroxide such as KOH in the presence of air or an oxidizing agent such as KNO3 to form dark green potassium manganate (K2MnO4).
Why are KMnO4 and K2Cr2O7 colored?
Potassium dichromate (K2Cr2O7) and potassium permanganate (KMnO4) are both colored compounds. The purple color of KMnO4 and the red-yellow color of K2Cr2O7 are due to the charge transfer spectra.
What is the best KMnO4 and K2Cr2O7 oxidant?
KMnO4 is a stronger oxidizing agent than k2Cr2O7 due to its higher reducing potential, as we know that the compound with the higher reducing potential performs as the best oxidizing agent. Here the reduction potential of KMnO4 is + 1.52V and K2Cr2O7 + 1.33V.
Which KMnO4 medium is a better oxidizing agent?
Why is K2Cr2O7 not self-indicating?
Potassium dichromate acts only as an oxidizing agent in an acidic environment. it cannot be used as an auto-indicator like KMnO4. in fact the reduction product (Cr3 +) is green, which prevents visual recognition of the end point by perceiving the color of the dichromate. An indicator is therefore essential for this titration.
Why is KMnO4 the best oxidant in an acidic environment?
(2) When reacting with an acid solution, more H + ions are used, which means that a lower pH value favors the reaction (Le Chateliers principle). The same is true for the reaction in neutral solution. For these reasons, KMnO4 is a stronger oxidizing agent in an acidic environment.
Why is KMnO4 acidified?
The acidified solution of potassium manganate (VII) oxidizes the olefin by breaking the carbon-carbon double bond and replacing it with two carbon monoxide double bonds. The products are called carbonyl compounds because they contain the carbonyl group C = O.
What oxidizes K2Cr2O7?
Description: Primary and secondary alcohols are oxidized to carboxylic acids or ketones by K2Cr2O7. Oxidation is physically observed through a color change due to the reduction of Cr6 + (yellow) to Cr3 + (blue). This demonstration also illustrates the chemistry behind the breath analysis test.
How much potassium permanganate dissolves in a liter of water at 20 ° C?
Can KMnO4 oxidize a tertiary alcohol?
You are right. Tertiary alcohols are easily removed from alkenes, after which KMNO4 reacts with the olefin to form syn-dihydroxylation.