Hypotonic meaning - How To Discuss
 Andrew Campbell
Andrew Campbell 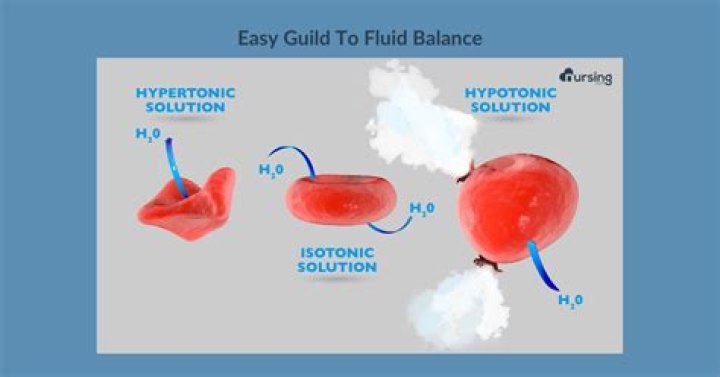
Hypotonic meaning
What is the difference between the hypotonic and hypertonic? Key Differences Between Hypertonic and Hypotonic Solution. A hypertonic solution has a high osmotic pressure and a low hypotension. The solute concentration is high in hypertensive patients and low in hypotensive patients. The solvent concentration is low in hypertensive patients and high in hypotonic patients. The cell dries up in a hypertonic solution, while the cell swells in a hypotonic solution.
What does hypotonic mean in biology?
Definition of hypotonic. Hypotonicity can mean several things in biology. The many definitions apply to both human biology and biochemistry. In humans or animals, hypotonic means that a muscle is less tight or less shaped than a normal reference model or compared to another muscle in the same human body.
Is D5W isotonic or hypotonic?
D5W is technically isotonic, but becomes hypotonic in the body, drawing fluid from the vascular system to the cells. This is because once D5W is ingested, the dextrose is metabolized leaving free water, which is hypotonic.
What is hypotonic in chemistry?
A hypotonic solution is a solution that has a lower concentration of solute compared to another solution. The solution cannot be hypotonic, isotonic, or hypertonic without a reference solution. Hypotonicity is a description of the content of solutes in one solution compared to another solution.
Why use hypotonic IV solutions?
Hypotonic solution: A solution that contains fewer dissolved particles (such as salt and other electrolytes) than in normal cells and blood. Hypotonic fluids are often used to administer intravenous fluids to patients to treat or prevent dehydration.
Is TPN hypertonic or hypotonic?
PN and PNP (total parenteral nutrition and partial parenteral nutrition) cause hypertension. However, I know that dextrose greater than or equal to 5% is considered hypertensive (D5NS, D5LR, D10). Albumin and highly concentrated electrolytes (potassium, magnesium), as well as blood products, are hypertonic.
![:brown_circle: :brown_circle:]() Which of these solutions is hypotonic?
Which of these solutions is hypotonic?
A hypotonic solution is a solution that contains less solute than a cell placed in it. If a cuvette with a concentration of NaCl is placed in a solution of distilled water, which is pure water with no solutes, the solution outside the cuvette consists of 100% water and 0% NaCl.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() What is the difference between hypotonic and hypertonic?
What is the difference between hypotonic and hypertonic?
The main difference between hypotonic and hypertonic drugs is that a hypotonic solution has a lower concentration of solutes than a cell while a hypertonic solution has a higher concentration of solutes than a cell… Osmosis is a process in which water molecules move from a high water potential to a low water potential through a semi-permeable membrane.
![:brown_circle: :brown_circle:]() What is the difference between the hypotonic and hypertonic solution
What is the difference between the hypotonic and hypertonic solution
A hypotonic solution is a solution with a lower osmotic pressure. The low osmotic pressure is the result of a low concentration of solutes. The key difference between hypotonic and hypertonic isotonic solutions is that isotonic solutions are solutions with the same osmotic pressure whereas hypotonic solutions are solutions with lower osmotic pressure and hypertonic solutions are solutions with high osmotic pressure.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() What does it mean to say a solution is hypertonic?
What does it mean to say a solution is hypertonic?
A hypertonic solution (pronounced HĪperTAWNik) is a solution with a higher solute concentration than the other specified solution (and therefore has a higher osmotic pressure than the other solution). So the solution with the lowest concentration is called hypotonic.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() What happens to a cell in a hypotonic solution?
What happens to a cell in a hypotonic solution?
A cell in a hypotonic solution can take up enough water to lyse or break the cell membrane, destroying the cell. Plant cells have a certain defense against this phenomenon, because their cell walls prevent cell breakdown.
What are some examples of hypotonic solutions?
Hypotonic solution Determination of the hypotonic solution. A hypotonic solution is a solution that has a lower concentration of solute compared to another solution. Examples of hypotonic solution. Large plants and fungi control the environment around their cells, ensuring that the environment is always a hypotonic solution for cells. Related terms from biology. test survey.
What does hypertonic mean
A hypertonic solution has a higher solute concentration than a cell and a higher osmotic pressure outside the cell than inside the cell. This will pull water out of the cell and force the cells to try to balance the osmotic pressure.
When would you give a hypertonic solution?
These solutions are very useful when cells have been poisoned with water, when they have been in a hypotonic environment for a long time and when they are swollen. Therefore, the introduction of a hypertonic solution causes cellular desiccation and will be beneficial to the cell.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() When is hypotonic solution used?
When is hypotonic solution used?
As for hypotonic solutions, it can be used to treat dehydration and hypernatremia or high sodium in the blood. Hypotonic solutions act on the body causing the cells to absorb water, causing edema. Since the solute is less in hypotonic solutions, the water from the solution migrates into the cell.
What are the effects of hypertonic environments?
When a cell enters a hypotonic environment (for example, pure water), the water diffuses into the cell and the cell begins to swell. This can of course cause an ■■■■■■■■■. Many bacteria have cell walls that protect them from such osmotic disruption (or osmotic lysis), creating a rigid barrier to the inflamed cell.
What does hypertonic and hypotonic and isotonic mean?
The key difference between hypotonic and hypertonic isotonic solutions is that isotonic solutions are solutions with the same osmotic pressure whereas hypotonic solutions are solutions with lower osmotic pressure and hypertonic solutions are solutions with high osmotic pressure.
What is the difference between the hypotonic and hypertonic fluid
Hypotonic solutions are used when the cell is dehydrated and fluids need to be replenished intracellularly. This occurs when patients develop diabetic ketoacidosis (DKA) or hyperosmolar hyperglycemia. Important: Be careful not to drain fluid from your circulation as you are trying to force extracellular fluid into the cell to rehydrate it.
![:brown_circle: :brown_circle:]() What are examples of hypotonic fluids?
What are examples of hypotonic fluids?
Examples of hypotonic fluids: NaCl (semi-concentrated saline) NaCl (one third of the saline), dextrose in water.
What is the difference between hypertonic hypotonic and Isotonic solutions?
The key difference between hypotonic and hypertonic isotonic solutions is that isotonic solutions are solutions with the same osmotic pressure whereas hypotonic solutions are solutions with lower osmotic pressure and hypertonic solutions are solutions with high osmotic pressure.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() What is the difference between the hypotonic and hypertonic cell
What is the difference between the hypotonic and hypertonic cell
Solutions contain less solutes and more solvents while hypertonic solutions contain more solutes and less solvents. Solutions cause the cell to swell, encouraging the movement of water through it, while hypertonic solutions compress the cell and draw water out of the cell.
What does hypertonic mean in science?
Hypertensive definition. In biology, hypertonicity refers to muscles that are more or less stiff than other muscles in the body, or stiffer than the "normal" comparative model.
What happens to cells in a hypotonic environment?
When exposed to a hypotonic environment, water enters the cell and the cell swells. If the water is not removed from the cell, the pressure will eventually exceed the maximum resistance of the cell and it will burst or collapse.
What is the solution that is hypotonic to a cell?
Substances dissolved in a hypotonic solution are also lower (in concentration) than in another solution. This means that there is more water in the hypotonic solution. For example, a cell in a hypotonic solution can cause water to enter the cell (diffuse). This, in turn, will cause the cell to swell.
What does hypotonic mean in medical terms?
The medical definition of hypotension. 1: Children with hypotension have low tone or tension. 2: It has lower osmotic pressure than the environment or liquid compared to hypotonic solution; Compare hypertensive sensation 2, isotonic sensation 1.
When do you use a hypotonic solution?
As for hypotonic solutions, it can be used to treat dehydration and hypernatremia or high sodium in the blood. Hypotonic solutions act on the body causing the cells to absorb water, causing edema.
Hypotonic meaning in biology
The many definitions apply to both human biology and biochemistry. In humans or animals, hypotonic means that a muscle has less tone or shape compared to a normal reference model or compared to another muscle in the same human body.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() What does isotonic mean
What does isotonic mean
Isotonic (a.) (Physiology, biochemistry) With osmotic pressure (tonicity) equal to the standard solution of aqueous solutions. Unlike the hypotonic and hypertensive. See hypertension.
What are the effects of isotonic?
Isotonic training causes a volume overload on the heart and increases oxygen consumption, heart rate, stroke volume, cardiac output and systolic blood pressure. As a result of reduced peripheral resistance during isotonic exercise, diastolic blood pressure may fall.
What does isotonic mean in relation to body fluids?
1. Isotonic. Of a fluid that exerts the same OSMOTIC PRESSURE as another, especially body fluids. Cells in the body, such as red blood cells, can be immersed in an isotonic solution without changing their shape.
What are examples of isotonic?
- Alcohol test. It's no secret that a glass of beer (about 30 g) affects them less physically than a glass of whiskey.
- Eliminate dehydration. The saline solution is water mixed with sodium chloride (NaCl).
- Isotonic exercises. When physical activity is considered isotonic, it has an ascending phase and a descending phase.
What does it mean to say that two solutions are isotonic?
ISOTONIC. In a general sense, two solutions are isotonic if they contain the same amount of solutes or solutes and therefore have the same osmotic pressure. However, isotonic solutions commonly used in the medical field are solutions that have the same concentration of solutes as cells in the human body.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() What does hypotonic mean in biology meaning
What does hypotonic mean in biology meaning
Definition of hypotonic. Hypotonicity can mean several things in biology. The many definitions apply to both human biology and biochemistry. In humans or animals, hypotonic means that a muscle is less tight or less shaped than a normal reference model or compared to another muscle in the same human body.
What does hypotonic mean in biology quizlet
What does hypotonic solution mean? A hypotonic solution has a lower concentration of solutes than another solution. In biology, a solution outside the cell is called hypotonic if it has a lower concentration of solute than the cytosol.
What is a hypotonic condition?
Hypotension means a decrease in muscle tone. On its own, it could be a condition known as benign congenital hypotension, or it could indicate another problem leading to a gradual loss of muscle tone, such as muscular dystrophy or cerebral palsy. This is usually found in early childhood. he or she is in prison.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() What is KCL in D5W?
What is KCL in D5W?
KCL in D5W (potassium chloride in 5% dextrose solution) represents fluid and electrolyte replacement and calorie intake used as a source of water and electrolytes. Side effects of KCL in D5W include fever, nausea, vomiting, abdominal pain, diarrhea and infection, redness, pain, or swelling at the injection site.
Is D5W and normal saline compatible?
Acetadot is hyperosmolar (2600 mOsm/L) and is compatible with 5% Dextrose (D5W), Saline (Sodium Chloride Injection, ½ NS), and Water for Injection (WFI). The total volume administered should be adjusted in patients weighing less than 40 kg and in patients requiring fluid restriction:.
Is D5 half NS hypertonic?
While D5 1/2 NS is hypertonic in the sac, when given intravenously, it becomes hypotonic because glucose is absorbed (almost instantaneously) by the body, so only 1/2 NS is absorbed into the vascular space. Improper administration of this fluid can cause edema and cell lysis.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() What is D5W IV solution?
What is D5W IV solution?
Description of liquid D5W (Dextrose 5% water) IV. 5% dextrose in water (D5W) increases total fluid volume and is also beneficial for rehydration and elimination. Type of solution Classification Content Mechanism of action. Dextrose provides calories. Indications Contraindications Nursing duties.
Why is d5w hypotonic
D5W is isotonic in solution, but becomes hypotonic in the body because the body uses dextrose. The osmolarity of the solution relative to the osmolarity of normal blood plasma determines whether the solution is iso, hyper, or hypotonic and determines the behavior of the fluid in the body.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() What are hypotonic solutions?
What are hypotonic solutions?
Hypotonic solution. A hypotonic solution is a solution that contains less solute than a cell placed in it. If a cuvette with a concentration of NaCl is placed in a solution of distilled water, which is pure water with no solutes, the solution outside the cuvette consists of 100% water and 0% NaCl.
Is d5w hypotonic or isotonic
D5W (5% dextrose in water) is an isotonic crystalloid liquid for intravenous administration with a serum osmolality of 252 mOsm/L. D5W is initially an isotonic solution and provides free water when dextrose is metabolized (turning it into a hypotonic solution) yielding ECF and ICF. will be expanded. It is administered to replace water and correct the rise in serum osmolality.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() What is D5W solution?
What is D5W solution?
The D5W solution is isotonic for dextrose metabolism. After dextrose metabolism, the D5W solution becomes hypotonic. It does not replace electrolytes. Contraindicated in renal failure or heart disease. Contraindicated in intracranial pressure or at risk of intracranial pressure. Do not mix dextrose with blood, as hemolysis is possible.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() Is lactated ringers hypotonic?
Is lactated ringers hypotonic?
Lactated Ringer's (RL) is an intravenous fluid commonly used in surgical procedures and has been shown to have a lower risk of hyponatremia, although it is a slightly hypotonic solution.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() What is hypertonic in chemistry?
What is hypertonic in chemistry?
A hypertonic solution is a solution with a higher osmotic pressure than another solution. In other words, a hypertonic solution is a solution in which the concentration or amount of solute particles on the outside of the membrane is greater than on the inside.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() Hypotonic meaning in science
Hypotonic meaning in science
Hypotonicity is a description of the content of solutes in one solution compared to another solution. It is used in biology to help scientists describe cells. Knowing the osmolarity (the concentration of a solution in the number of solutes per liter) of different solutions can show scientists how water gradients and solute gradients form.
What is hypotonic in chemistry quizlet
A solution with the same concentration of water and solutes as the cell, which allows the cell to maintain its normal shape because there is no pure movement of water. What happens to a cell in a hypertonic solution? The effect of a hypertonic solution on animal and plant cells.
What is hypotonic in chemistry terms
A hypotonic solution is a solution that has a lower concentration of solute compared to another solution. The solution cannot be hypotonic, isotonic, or hypertonic without a reference solution. Hypotonicity is a description of the content of solutes in one solution compared to another solution. It is used in biology to help scientists describe cells.
What is hypertonic and hypotonic?
A hypotonic solution is a solution containing low concentrations of a solute and a hypertonic solution is a solution containing high concentrations of a solute. Thus, this is the key difference between hypotonic and hypertensive. Furthermore, a hypotonic solution has a high water potential and a hypertonic solution has a low water potential.
![:brown_circle: :brown_circle:]() What is hypotonic in chemistry examples
What is hypotonic in chemistry examples
A hypotonic solution is a classic example of water. However, this will also be subjective to the solution being compared. Purified distilled water is a solvent and is always hypotonic for an aqueous solution of a solute containing any amount of a solute.
![:brown_circle: :brown_circle:]() What is hypotonic in chemistry for dummies
What is hypotonic in chemistry for dummies
Hypotonic solutions are the exact opposite of hypertonic solutions. Cells usually have a lower water concentration than outside the cell. As a result, the solutes outside the cell are lower than the solutes inside the cell.
What does the Hypo mean in biology terms?
Hiccups - The prefix means weak, below, below, below or below normal, as in hypoglycemia (low blood sugar) and undersensitivity (hypersensitivity). The opposite of Hypo is hyper.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() How do tell if hypertonic or hypotonic?
How do tell if hypertonic or hypotonic?
Key Differences Between Hypertonic and Hypotonic Solution. A hypertonic solution has a high osmotic pressure while a hypotonic solution has a low osmotic pressure. The concentration of a solute in a hypertonic solution is higher than in a hypotonic liquid. The solvent concentration is low in hypertonic and high in hypotonic.
Hypertonic isotonic hypotonic meaning
Hypotonic: has a lower concentration of fluid, sugar and salt than blood Isotonic: has a comparable concentration of fluid, sugar and salt in the blood Hypertonic: has a higher concentration of fluid, sugar and salt than blood Each drink has different Benefits for you Guide your education .
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() What does hypertonic mean?
What does hypertonic mean?
Hypertensive definition. In biology, hypertonicity refers to muscles that are more or less stiff than other muscles in the body, or stiffer than the "normal" comparative model.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() Facilitated diffusion meaning
Facilitated diffusion meaning
Facilitated diffusion is a form of facilitated transport in which the passive movement of molecules along a concentration gradient is guided by the presence of another molecule, generally an integral membrane protein that forms a pore or channel.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() What is the difference between facilitated and diffusion?
What is the difference between facilitated and diffusion?
In fact, active transport involves particles moving against their concentration gradient, while facilitated diffusion moves particles along a concentration gradient.
What are three facts about Facilitated diffusion?
The main factors influencing the facilitated diffusion process are: Temperature As temperature increases, the movement of molecules increases due to the increase in energy. Concentration The movement of molecules takes place from the region of the highest concentration to the region of the lowest concentration. Diffusion Distance The diffusion rate is greater in a smaller distance than in a large one.
What is required for a facilitated diffusion to occur?
Simple diffusion does not require energy: facilitated diffusion requires a source of ATP. Simple diffusion can only move material in the direction of the concentration gradient. Facilitated diffusion moves materials with and against a concentration gradient.
![:brown_circle: :brown_circle:]() Hypotonic meaning osmosis
Hypotonic meaning osmosis
Osmosis: A process in which a liquid moves through a semi-permeable membrane. Semi-permeable membrane: A membrane that allows solvent to pass through but does not contain solutes. Let them now determine what the tonicity of the solutions can be: Hypotonic (definition): little solute, high solvent content.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() What are the examples of hypertonic solution?
What are the examples of hypertonic solution?
A hypertonic solution is a solution in which the concentration of a solute is higher than in another solution. An example of a hypertonic solution is the interior of an erythrocyte versus the concentration of a solute in fresh water.
Why does a hypotonic solution causes a cell to burst?
When you have a HYPOTON SOLUTION it means that there are more solutes in the cell than in the solution so the water of osmosis enters the cell to reach equal concentrations. This causes the cell to swell and burst. This means that the cell is hypertonic to the solution, when the solution is hypotonic to the cell.
Bio hypotonic meaning
The definition of hypotonic hypotension can have several meanings in biology. The many definitions apply to both human biology and biochemistry. In humans or animals, hypotonic means that a muscle is less tight or less shaped than a normal reference model or compared to another muscle in the same human body.
![:brown_circle: :brown_circle:]() What is isotonic,hypertonic and hypotonic?
What is isotonic,hypertonic and hypotonic?
A hypertonic solution is a solution that contains more solute, a hypotonic solution contains more water, and an isotonic solution contains equal amounts of solute and water. A hypertonic, hypotonic or isotonic solution can determine what happens to the cell.