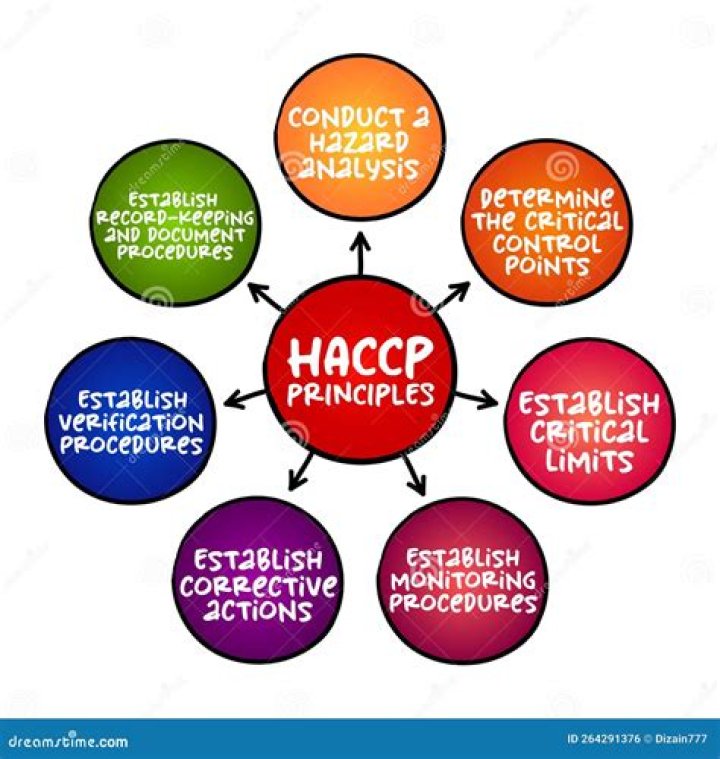
Hofbrincl - How To Discuss
 Andrew Campbell
Andrew Campbell 
Hofbrincl
What is FBrINCl (Chemistry)? 3
I know what that means, I mean what do you do with it? : /
I am studying for a science exam and I have not taken this class yet.
Help would be appreciated, thanks :)
It only helps you remember the diatomic molecules. In general, if you have one of these equations or equations, molar m needs to be doubled in terms of the number of extra atoms in the molecule.
All are diatomic molecules.
You represent
H = hydrogen
O = oxygen
F = flow
Bur = bromine
I = iodine
N = nitrogen
Cl = chlorine
The acronym FBrINCl stands for 7 chemical elements in the form of diatomic molecules. This means that under neutral conditions (standard temperature and pressure) they are present in pairs instead of individual atoms (ie HH, OO, FF, etc.). This tendency is the result of your ■■■■ structure.
For your information: Sulr exists as the 8-atom ring of Sulr in neutral condition! (It is polyatomic rather than diatomic).
Used to reduce hydrogen. Increase and decrease of hydrogen. H + means proton, an acid ion. Oxygen. The addition of oxygen is oxidation. It is used for breathing. There are fluorine, chlorine, bromine and iodine halides. They are present with acids that make hydrogen. The acid turns into salt and water. Nitrogen is abundant in the atmosphere. Forms a hydrogen base called ammonia.