Co2 Bond Angle - How To Discuss
 Daniel Cobb
Daniel Cobb 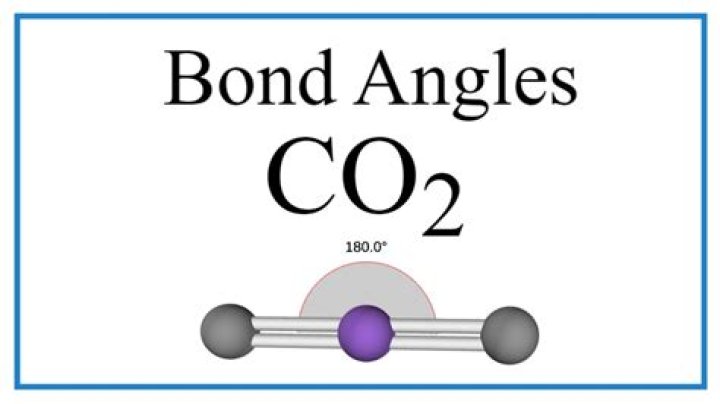
Co2 Bond Angle
Why is the bond angle at carbon dioxide, CO2, 180 degrees?
And, I'm sure you're wrong. Cinema bonds and pie bonds are required for double bonds.
Two theories work to predict the shape of a molecule.
(1) Valence electron pair violation theory (RPECV):
The arrangement of atoms around the central atom depends on the total number of valence electron pairs around the central atom, including bonded and non-bound pairs.
Valence electron pairs adopt a system that determines the distance between two pairs, as pairs of electrons have a strong tendency to repel each other.
Inbound electron pairs (isolated pairs) take more and more than electron pairs. Therefore, the retreat between two pairs of solitary electrons is greater than the retreat between two pairs of bound electrons.
The invasive forces between pairs of electrons can be summarized as follows:
Isolated pair isolated pair> isolated connection pair> connection pair connection pair.
(2) Hybridization Theory:
Including hybrids.
(a) All electron pairs are undivided (isolated pairs).
(b) Binding pairs of electrons into single bonds (sigma bonds)
(c) One and only one pair of electrons in a double or triple bond (excluding sigma bonds, pie bonds)
In terms of molecular geometry, double bonds behave like single bonds. In other words, the pair of extra electrons in a double or triple bond has no effect on the geometry of the molecule.
Molecular geometry is determined by the pairs of electrons in the ■■■■ hybrid for the central atom. In hybrids, these electron pairs can be bond pairs or single pairs. According to VSEPR theory, these electron pairs are based on a maximum and determine the shape of a polyatomic molecule or ion.
Now apply these ideas to your molecules.
CO2:
O = C = O.
Carbon has 4 valence electrons and all these electrons are used to bind to two O atoms (it has 6 valence electrons) because oxygen needs two electrons to make it octet, so both oxygen Each of them divides two electrons with carbon, so all three atoms complete the octet in terms of distribution ..... The central atom of C does not have isolated pairs. There, in the CO2 molecule, the bond pairs are repulsed. The parental distance between the couple can be obtained from a 180 degree bond angle. There, the molecule is linear. You can also draw conclusions from this statement. The type of hybridization is C ......
Generalization: If the hybridization type of the central atom is SP, the shape of the molecule is linear.
Ah, I missed something ... that's a better explanation ... you are right about the formation of double bonds, there are sigma bonds and pie bonds ...
For this reason. In CO2, the value of oxygen is 2. Since carbon has a valence of 4, it must form 4 bonds with two oxygen. This means that carbon forms two double bonds with oxygen. So there must be two sigma bonds and two pie bonds.
Co2. Connection angle
Co2 Bond Angle
Co2 Bond Angle
Why Carbon Dioxide, CO2, Bond Angle at 180 Degrees? 3
Please explain and include what is verbal and where sigma and pi link is, I need to understand it better.
updateAlso, try to go into detail about double bonding.
And, I'm sure you're wrong. Cinema bonds and PI bonds are required for double bonds.
Two theories are useful in predicting the shape of a molecule.
(1) Theory of S-Valence Electron Pair Repulsion (VSEPR):
The arrangement of atoms around the central atom depends on the total number of pairs of valence electrons around the central atom, including bound and non-bound pairs.
The valence electron pair adopts an arrangement that minimizes the distance between the two pairs, as the pairs of electrons have a strong tendency to repel each other.
Non-bound electron pairs (isolated pairs) take up more and more of the electron pairs. Therefore, the repetition between two solitary pairs of electrons is greater than the repetition between two pairs of bound electrons.
The concentration forces between pairs of electrons can be summarized as follows:
Isolated pair isolated pair> isolated connection pair> connection pair connection pair.
(2) Hybridization Theory:
Including hybrids
(a) All electron pairs are undivided (isolated pairs).
(b) The union of electron pairs into single bonds (sigma bonds)
(c) One and only one pair of electrons in double or triple bonds (excluding sigma bonds, pie bonds).
In terms of molecular geometry, double bonds behave like single bonds. In other words, the pair of extra electrons in a double or triple bond has no effect on the geometry of the molecule.
Molecular geometry is determined by the pairs of electrons in the ■■■■ hybrid for the central atom. In hybrids, these electron pairs can be bond pairs or isolated pairs. According to VSEPR theory, these electron pairs are maximally oriented and determine the shape of the polyatomic molecule or ion.
Now apply these ideas to your molecules.
CO2:
O = C = O
Carbon has 4 valence electrons and all these electrons are used to bind to two O atoms (it contains 6 valence electrons) because oxygen needs two electrons to make its octet, so both oxygen. Each of them shares two electrons with the carbon, so all three atoms complete the octet in terms of distribution ... The central atom of C does not have isolated pairs. There, in the CO2 molecule, the pair of bonds is pushed back. The parent distance between the pair can be obtained from a 180 degree bond angle. There, the molecule is linear. You can also draw conclusions from this statement. Hybridization type C is sp ......
Generalization: If the hybridization type of the central atom is sp, then the shape of the molecule is linear.
Oh I missed something ... Here's a better explanation ... You're right about double bond formation, sigma bonds and pie bonds ...
For this reason. In CO2, the value of oxygen is 2. So it should make two bonds. Since carbon has a balance of 4, it must form 4 bonds with two oxygen. This means that carbon forms two double bonds with oxygen. So there should be two sigma bonds and two pie bonds.
This page can help you.
D:
Why Carbon Dioxide, CO2, Bond Angle at 180 Degrees?
Please explain and include what is verbal and where sigma and pi link is, I need to understand it better.
C) Because the oxygen molecule has more single pairs of electrons than the carbon molecule, the hydrogen molecule is pushed back further by the negative boost due to the electrons, causing a different bond angle. Imagine the linear structure of a CO2 molecule due to the harmony of the bonds. The vertical and horizontal balance is due to the fact that no electrons are unequally distributed. .. .. O = C = O .. .. while horizontal H2O is disproportionate to observation. When electrons of hydrogen molecules combine with oxygen molecules, they do not have extra electrons to balance the polarity. The positive nature of the hydrogen nucleus repels the rest of the electrons from oxygen, which forms the angle of their bond ... O H H
Double bonds do not have sigma and pie bonds. This is a hybrid of tse bond called sp hybrid bond.



