Calcium Electronegativity - How To Discuss
 Amelia Brooks
Amelia Brooks 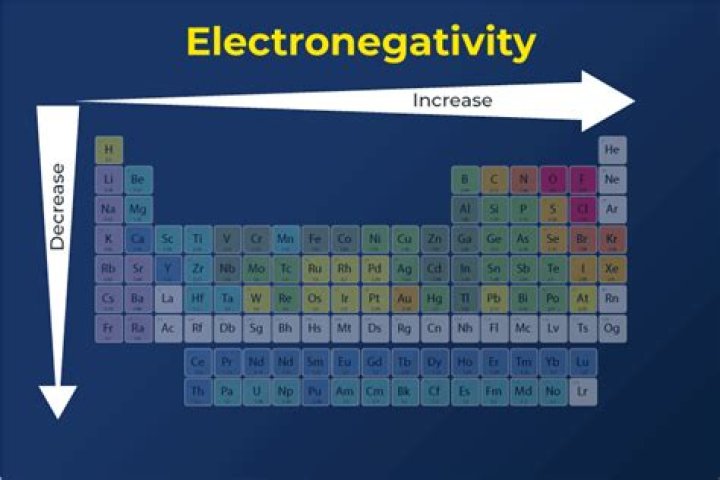
Calcium Electronegativity
Calcium has high or low electronegativity
So the lowest value will be on the bottom left - of these atoms, i.e. cesium. Electronegativity increases over time. Electronegativity increases when you join a group. Calcium is higher in the group than barium, so it has higher electronegativity.
What is the electronegativity of calcium?
1Which elements also have greater electronegativity?
Of the main group elements, fluorine has the highest electronegativity (EN = 4.0) and cesium the lowest (EN = 0.79). This indicates that fluorine has a high tendency to absorb electrons from other elements with lower electronegativity.
Also note that potassium has high or low electronegativity?
Sodium has a higher electronegativity than potassium. Sodium and potassium belong to the same family. The farther the electron density is from the positive nucleus, the lower the electronegativity, the lower the positive charge on the nucleus, the lower the electronegativity.
How does electronegativity compare?
Find bonds with electronegativity. Find the difference in electronegativity between the two atoms. When two atoms are bonded together, the difference between their electronegativities can tell us the properties of their bond. Subtract the lower electronegativity from the higher to find the difference.
What is the ionization energy of nitrogen?
The ionization energy of molecular nitrogen is 1503 kJ mol 1 l and that of atomic nitrogen 1402 kJ mol 1 1. Also here the energy of the electrons of the molecular nitrogen is lower than that of the electrons of the individual atoms, therefore the molecule is tied up.
Where is the highest electronegativity?
Electronegativity increases in groups from bottom to top and from left to right over time. Therefore, fluorine is the most electronegative element, while francium is one of the least electronegative.
Is HCL polar or non-polar?
HCl is a polar molecule because chlorine has a higher electronegativity than hydrogen. It then attracts electrons to pass more time, which gives it a negative charge and hydrogen a positive charge.
How do you know if Br2 is polar or non-polar?
Which element has the least electronegativity?
Francio Why is fluorine more electronegative?
Fluorine is the most electronegative element as it has 5 electrons in the 2P shell. The optimal electron configuration of the 2P orbital contains 6 electrons. Because fluorine comes so close to the ideal electronic configuration, the electrons are kept very close to the nucleus.
What is the ionization energy of calcium?
Ionization energies and electron affinity The electron affinity for calcium is 2.37 kJ mol1.
Which element has the highest ionization energy?
This trend shows that cesium has the lowest ionization energy and fluorine has the highest ionization energy (excluding helium and neon).
What are the four most electronegative elements?
On this scale, neon has the highest electronegativity of all elements, followed by fluorine, helium and oxygen.
What is called high electronegativity?
Electronegativity is a measure of an atom’s tendency to attract a pair of bonding electrons. The Pauling scale is the most used. Fluorine (the most electronegative element) is given a value of 4.0 and the values rise to Cesium and Francium, which are the least electronegative at 0.7.
What is electronegativity in simple terms?
Electronegativity is a measure of how strongly atoms attract bonding electrons. Below is a periodic table of electronegativity: the lighter the green color, the higher the electronegativity. Gray means that no value is known.
What are the causes of high electronegativity?
Electronegativity increases as you move from left to right on the periodic table. This is due to a higher charge on the nucleus, which makes the electron bond pairs very attracted to the atoms that are more directly on the periodic table. Fluorine is the most electronegative element.
What do the electronegativity values say?
Electronegativity is the tendency of a bonded atom to attract shared electrons. The greater the electronegativity, the greater the attraction. Differences in electronegativity between atoms can indicate the nature of the bond.
What is the most electropositive element?
Alkali metals are the most electropositive. Cesium is the most electropositive of all stable elements. Francium, although unstable, is theoretically the most electropositive element. Electropositivity increases groups and decreases over periods (left to right) in the periodic table.
What is an example of electronegativity?
Hydrogen has an electronegativity of 2.0 while oxygen has an electronegativity of 3.5. The difference in electronegativity is 1.5, which means that water is a polar covalent molecule. This means that the electrons are clearly attracted to the more electronegative element, but the atoms are not ionized.