Bf3 Polar Or Nonpolar - How To Discuss
 Amelia Brooks
Amelia Brooks 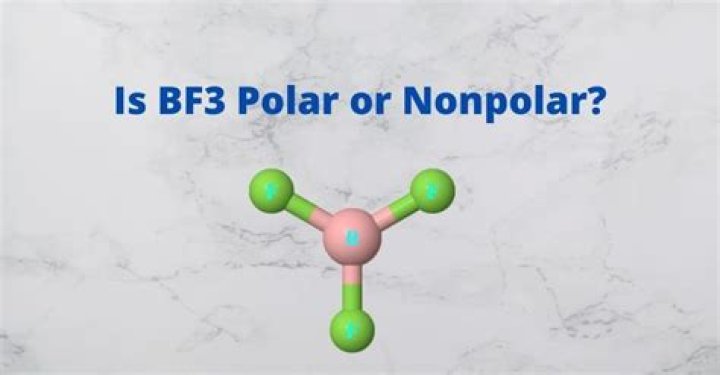
Bf3 Polar Or Nonpolar
Why is boron trifluoride (BF3) a non-polar factor?
Flat tribunal molecules have no opposite bonds. Each BF title is 120 degrees apart. This high degree of equilibrium means that there is no dopole moment in BF3, so the molecule is non-polar.
This page can help you.
Come back:
Why is boron trifluoride (BF3) a non-polar factor?
Whenever I draw a Lewis structure and an arrow pointing to the most electric atom in the bond, there is a doppel moment that becomes clear to me. The BF3 is a similar plane and I see that 2 opposing BF bonds have been canceled, but the third one creates a liquid dupole moment because it has no other bond ...
Bf3 Polar Or Nonpolar
Bf3 Polar Or Nonpolar
Why is Boron Trifluoride (BF3) a non-polar molecule? 3
Whenever I draw Lewis's skeleton and draw an arrow pointing to the most electronegative atom in the bond, there is a clear dupole moment for me. The BF3 is a triangular plane and I can see two opposing BF bonds being canceled, but the third one creates a liquid dupole moment because there is no other bond to cancel. Can anyone tell me why the molecule is actually non-polar?
Triangular planar molecules have no opposing bonds. Each LF link is another two to 120 degrees. This advanced synchronization means that there is no dopole moment in BF3, so the molecule is non-polar.
This page can help you.
D:
Why is Boron Trifluoride (BF3) a non-polar molecule?
Whenever I draw Lewis's skeleton and draw an arrow pointing to the most electronegative atom in the bond, there is a clear dupole moment for me. The BF3 is a triangle plane and I can see that 2 opposing BF bonds have been canceled, but the third one creates a liquid dupole moment because it has no other bonds ...
Bf3. Molecule
Bf3 Polar Or Nonpolar
Bf3 Polar Or Nonpolar
Think of the three dimensional structure of these molecules. HCl and BF bonds are polar. Unlike HCl, however, BF3 is perfectly symmetrical. This coordination will break all polar bonds and leave the global molecule without permanent polarity.
Lewis Bf3 structure
Molecular shapes do not have two opposite BF bonds, the third points in a different direction, the three BF bonds point in the vertical of an equilateral triangle.
...
For good reason, they have to cancel each other out. You can also test it with vectors.