Acido Sulfhidrico Formula - How To Discuss
 Alexander Torres
Alexander Torres 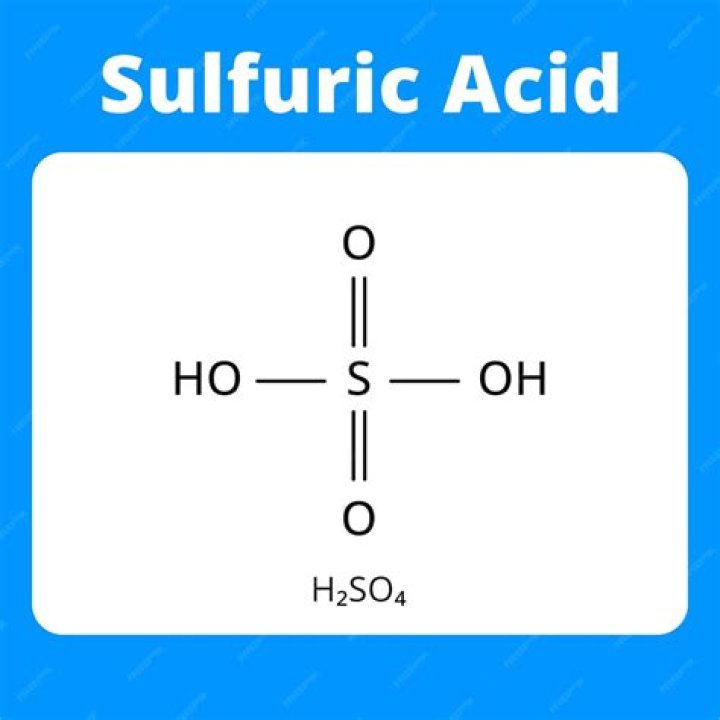
Acido Sulfhidrico Formula
The formula and equation for the formation of sulfuric acid?
Sulfuric acid consists of two hydrogen atoms and one sulfur atom. According to the acidity of water, its substance is intoxicating, lacks oxygen and, in the case of sulfur, it leads to a low state of oxidation, which is 2.
It is very simple to form, consists of two hydrogen atoms (hydrogen molecules), has a blue atom and the result is described above.
Peace, Greeting!
Hydrogenate with sulfur: H2S
S + H2 = H2S
Hydrogen sulfide is a solution of hydrogen sulfide (H2S) in water.
S + HH x SHH
H2SO
H2S formula (wonderful fragrance)
Always form Q mixture of sulfur with hydrogen.
H2 + S> H2S
Acido Sulfhidrico Formula
Acido Sulfhidrico Formula
H2S (hydrogen sulfide)
2H2 + S2 uce 2H2S
Formula H2SO4 is a corrosive, highly viscous and colorless liquid with a density of 1.85. Its settling point is 10.36 C, which is the boiling point of 340 3 C when dissolved in water in proportion. Mixing sulfuric acid in water will release a large number of calories. If the mixture is not stirred, the added water may overheat above the boiling point and a sudden increase in heat may cause the acid to escape from the can. Concentrated acids kill skin and flesh and can cause blindness if visible to the ■■■■■ eye. Washing the acid with plenty of water is the best treatment in case of an accident. Despite the great dangers of dealing with negligence, sulfuric acid has gained great commercial importance over the years. Eyebrows produce large amounts of it, warming their natural body at high temperatures and dissolving sulfur trioxide in water. In the fifteenth century, a method of extracting acid by extracting hydrated iron sulfate (or ferrous vitreous) from sand was developed. In 1740, commercial use of acid was started by burning sulfur and isotonics in a kettle suspended in a large glass ball partially filled with water.
Property
Sulfuric acid is a tartaric acid, which easily dissolves in aqueous solution of hydrogen ion (H +) and sulfate ion (SO42). Each molecule uses two H + ions, which means that sulfuric acid is the primary. Its aqueous solution has all the properties of acid: it is bitter, conducts electricity, neutralizes alkalis and inert metals, releases hydrogen gas. A salt containing the SO4 sulfate group and an acid salt containing the HSO hydrogen group can produce sulfuric acid.
Concentrated sulfuric acid, formerly known as vitreous oil, is an important source of dissolution. In this case, it works so well that it removes water and burns, allen, sugar and paper. Due to its drying properties, it is used in the manufacture of ether, nitroglycerin and dyes. For example, when heated, sulfuric acid concentrated as an oxidizing agent may be able to dissolve non-copper metals such as copper, mercury and lead, producing metal sulfate, which dissolves sulfur and water. Do
In the 19th century, the German chemist Justin von Leigh discovered that sulfuric acid increased the amount of phosphorus in plants by increasing the amount of soil. As a result of this invention, the commercial production of this acid increased, thus improving the manufacturing process.
Preparation
There are currently two methods used to obtain sulfuric acid. Both require the use of sulfur oxide in the initial stage, which is obtained by burning iron pyrite, FES2 or sulfur in the air. In the first process, called the Tin Chamber Matteo, the reaction reaches C in a large brick tower made of sesame. In this tower, sulfur dioxide reacts with gas, air, water vapor and nitrogen oxides, and produces sulfuric acid in the form of fine droplets falling on the floor of a room. Almost all nitrogen oxides emitted from sewage gas are left in the recycling chamber. In this form, sulfuric acid and labeled acid contain only 62 to 70% of H2SO4, the rest is water. Currently, about 20% of sulfuric acid is produced by the lead chamber process, meaning that this ratio is declining.
The extraction method, a contact method that was commercially introduced around 1900, is based on the oxidation of sulfur dioxide by a catalyst called sulfur trioxide, SO3. Finely twisted platinum, the most effective catalyst, has two disadvantages: it is very expensive and, in addition, some dirt, usually sulfur dioxide, poisons it and reduces its activity. Is. The sulfuric acid machine uses two catalysts: a primer, which is stronger but less efficient, such as iron oxide or vanoxide, which is processed and then further processed or melted. At 400 ° C, the conversion of dioxide to sulfur trioxide is almost complete. Trioxide is soluble in concentrated sulfuric acid and at the same time maintains the water flow concentration at the selected level, usually 95%. By reducing the flow of water, you get more SO3 products than in the H2SO4 formula. For organic chemistry reactions, this product is called mante sulfuric acid, oleum or nordhassan acid.
Auction
The amount of sulfuric acid varies so much that the auction quantity gives an estimate of the overall index of industrial activity. For example, the largest annual production of sulfuric acid in the United States was 29 million tons in the early 1970s, equivalent to a daily intake of 1/3 kg per person per year. Sulfuric acid is mainly used to make fertilizers, both super pit fertilizers and low ammonium fertilizers. Tamim is used to make organic, paint and lubricants and viscose, as well as to refine petroleum and processed metals. One of the consumer products containing sulfuric acid is the lead acid battery used in automobiles.
Acido Sulfhidrico Formula
Acido Sulfhidrico Formula
The formula and equation for the formation of sulfuric acid? 3
Hydrogen sulfide consists of two hydrogen atoms and one sulfur atom. According to hydrochloric acid, the substance is intoxicating, it lacks oxygen and, in the case of sulfur, it leads to a low oxidation state, which is 2.
Its formation is very simple, as it consists of two hydrogen atoms (hydrogen molecules) which have a blue atom, as described above.
Definitions!
The alternative current to hydrogen sulfide is: H2S
S + H2 = H2S
H2S formula (pleasant smell)
Always form q of sulfur with hydrogen.
H2 + S> H2S
Formula H2SO4 is a corrosive, highly viscous and colorless liquid with a relative density of 1.85. It has a settling point of 10.36 ° C and a boiling point of 340 ° C when dissolved in water. When sulfuric acid is added to water, a great deal of heat is released. If the mixing is incomplete, the added water can heat up above the boiling point and a sudden increase in heat can cause acid to escape from the container. Concentrated acids damage the skin and flesh and can cause blindness if they get into the eyes. The best treatment in case of an accident is to wash the acid with a large amount of water. Despite the serious risks of dealing with negligence, sulfuric acid has been of great economic importance for many years. Fry produces large quantities of it, warming its natural body to a higher temperature and dissolving sulfur trioxide in water. By the 15th century, a method of extracting acid was developed by distilling hydrated iron sulfate (or ferrous vitreous) with sand. In 1740, acid began to be used commercially by burning sulfur and sodium nitrate in a kettle suspended in a large glass ball partially filled with water.
Properties (edit)
Sulfuric acid is a turbid acid, meaning it dissolves easily in aqueous solution in hydrogen ions (H +) and sulfate ions (SO42). Each molecule uses two H + ions, which means that sulfuric acid is the basic. Its aqueous solution has all the properties of acids: they are bitter, conduct electricity, neutralize alkalis and degrade active metals, emit hydrogen gas. With sulfonic acids you can sell acids with Solo SO4 group and Solo HSO4 hydrogen groups.
Concentrated sulfuric acid, formerly called vitriol oil, is an important source of dissolution. It works so strongly that it drains out water and thus burns grain, grain, sugar and paper. Due to its drying properties, it is used in the manufacture of ether, nitroglycerin and dyes. When heated, sulfuric acid concentrated as an oxidizing agent, for example, can dissolve slightly reactive metals such as copper, mercury and lead, which produces metal sulfates, which are sulfur. And dissolve water.
In the 19th century, the German chemist Justice von Leigh discovered that adding sulfuric acid to the soil increases the amount of phosphorus available to plants. As a result of this invention, the commercial production of this acid increased, thus improving the manufacturing counters.
Manufacturing
There are currently two methods used to obtain sulfuric acid. Both initially require the use of sulfur oxide, which is obtained by burning iron pyrite, FeS2 or sulfur in the air. In the first process, called the tin chamber method, the AC response is increased in large towers made of tin bricks. The tower reacts with sulfur dioxide gas, air, water vapor, and nitrogen oxides, producing sulfuric acid in the form of fine droplets that fall to the floor of the room. Almost all nitrogen oxides emitted from sewage gases are sent to the chamber for reuse. In this form, sulfuric acid and labeled acid contain only 62 to 70% of H2SO4, the rest being water. About 20% of sulfuric acid is currently produced by local climate change, a proportion that is declining.
The process of silk fabrication, a cohesive process that began commercially around 1900, is based on the oxidation of sulfur dioxide to sulfur trioxide, SO3, a balancer. Finely divided platinum, the most efficient catalyst, has two disadvantages: it is very expensive and, in addition, some of the impurities in sulfur dioxide usually poison it and reduce its activity. ۔ Sulfuric acid mitochondria use two catalysts: a primer, which is stronger but less efficient, such as iron oxide or vanoxide, than the reaction, and then further processing or reducing the barrier. At 400 ° C, the conversion of dioxide to sulfur trioxide is almost complete. Trixide dissolves in concentrated sulfuric acid during flow with water, which maintains a concentration at a selective level, usually 95%. By reducing the flow of water, you get a product with more SO3 than the H2SO4 formula. This product, called mante sulfuric acid, oleum or nordhausen acid, is required for reactions in organic chemistry.
Auction
The amount of sulfuric acid varies so much that its auction volume provides an estimate of the overall index of industrial activity. In the United States, for example, the annual production of sulfuric acid was 29 million tons in the early 1970's, equivalent to 1/3 kg per person per day per year. Sulfuric acid is mainly used to make fertilizers, both super pit fertilizer and low ammonium fertilizer. Tamé n is used to produce organic, paint and lubricants, and viscose, as well as to refine petroleum and to process metals. One of the few consumer products that contains sulfuric acid is lead acid batteries, which are used in automobiles.