A 9.50 By Mass Solution Of Acetone
 Isabella Browning
Isabella Browning 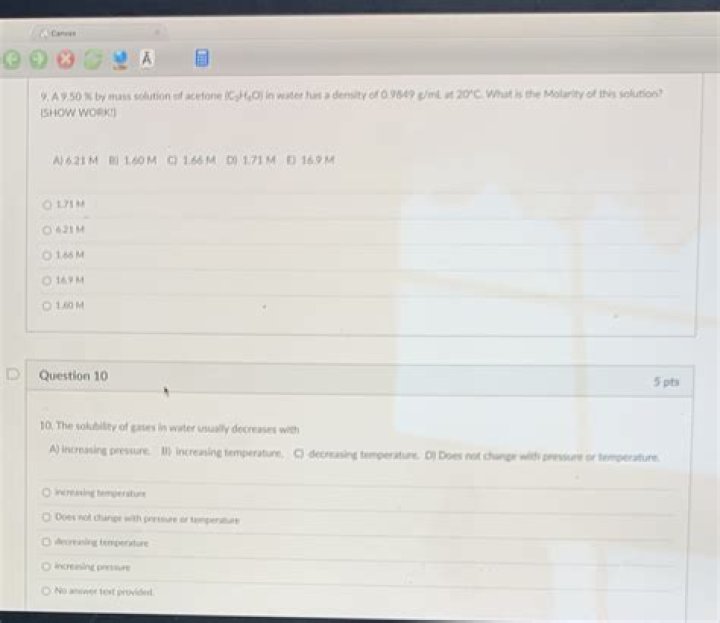
A 9.50 By Mass Solution Of Acetone
Does 9.50% solution of acetone (C3H6O) in water have a density of 0.9849 g / mL at 20 C per m2? 3
What is the molarity of the solution?
A) 1.61 million
B) 1.66 million
C) 1.71 million
D) 16.9 million
Density = meter / volume
m = density x volume
m = 0.9849 g / ml x 1000 ml
m = 984.9 grams
m acetone = 0.095 x 984.9 g
M Easton = 93.57
Acetone = 93.57 / 58.08 g / mol
Acetone = 1.61 estone / L = 1.61 m
Cute)



